Investigating the Association and Causality Between Hyperuricemia and Peripheral Atherosclerosis: A Study Based on Mendelian Randomization and Bioinformatics Analysis
DOI:
https://doi.org/10.71321/4w6y6229Keywords:
Hyperuricemia, Atherosclerosis, Mendelian randomization, Bioinformatics, Hub targets, Immune infiltrationAbstract
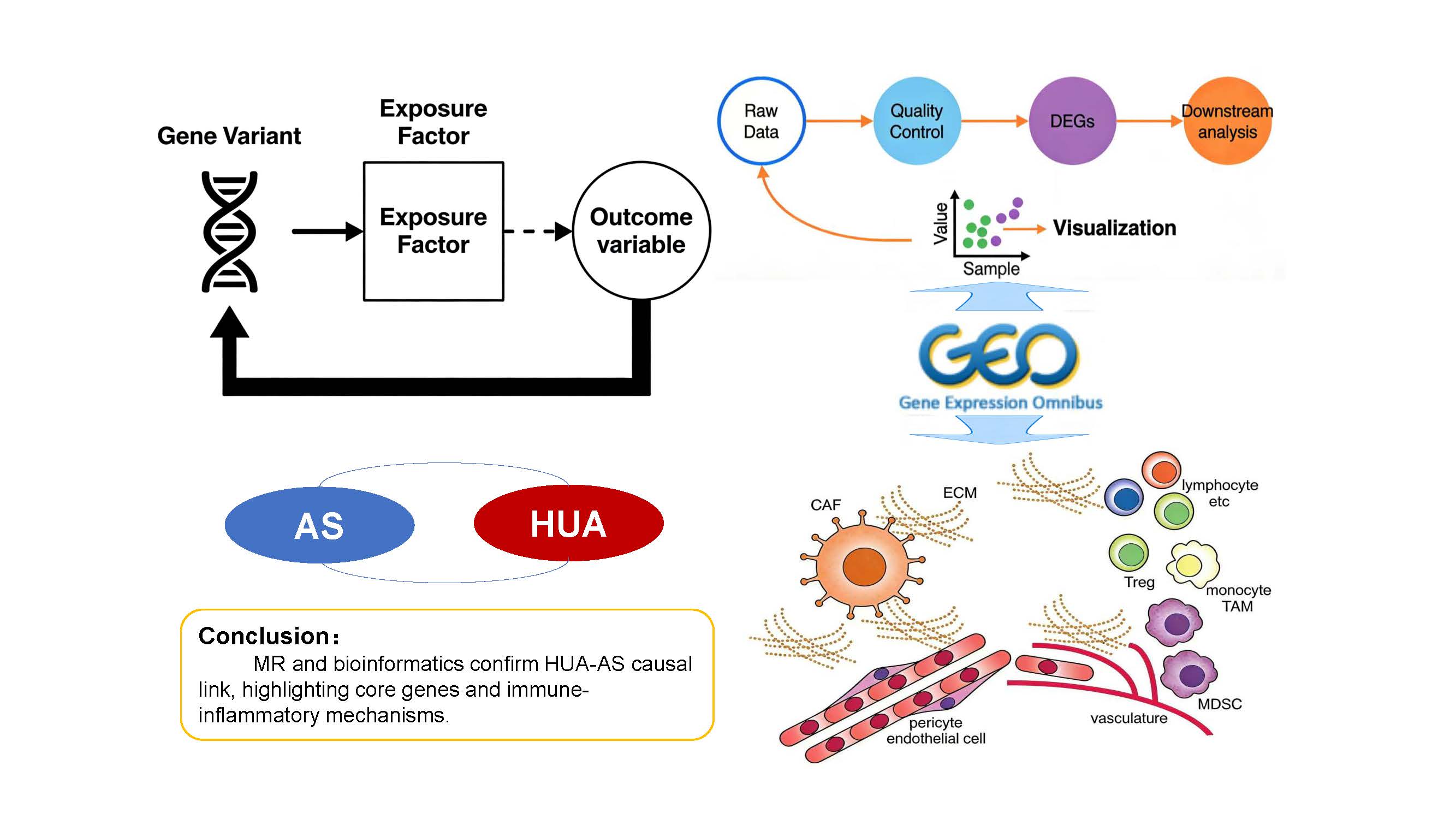
Objective: To investigate the genetic causal association and shared molecular mechanisms between hyperuricemia (HUA) and peripheral atherosclerosis using Mendelian randomization (MR) and bioinformatics approaches, providing a theoretical basis for early disease screening and intervention.
Methods: An MR strategy was employed. Based on Genome-Wide Association Study (GWAS) datasets, the causal relationship between HUA and peripheral atherosclerosis was assessed through instrumental variable selection, multi-model MR analysis (including Inverse-Variance Weighted, IVW), and sensitivity analysis validation. Simultaneously, relevant datasets from the GEO database were utilized to identify differentially expressed genes (DEGs) common to both diseases. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were performed, and a Protein-Protein Interaction (PPI) network was constructed to identify hub genes. Immune infiltration characteristics were analyzed using CIBERSORT.
Results: A significant positive causal association was found between hyperuricemia and atherosclerosis (OR=1.296, 95% CI: 1.112-1.512, P=0.0009). Bioinformatics analysis identified 133 intersecting DEGs, which were enriched in immune-inflammatory-related functions and pathways. Hub genes screened from the PPI network included IL1B, CD86, and CSF1R. Immune infiltration analysis revealed characteristic remodeling of the immune microenvironment in both groups.
Conclusion: A significant positive genetic causal association exists between HUA and peripheral atherosclerosis. The shared pathogenic mechanisms may involve the aforementioned hub genes, activation of immune-inflammatory pathways, and abnormal immune infiltration. The findings provide a theoretical framework and potential experimental targets for early risk prediction and targeted intervention in HUA complicated with peripheral atherosclerosis.
References
[1] Ouyang Y, Sun L, Yu D, Li Q, Du S, Wang X, et al. (2025). Exogenous chemical exposures and metabolic disruptions in hyperuricemia: a multi-omics mediation study. Environ Int, 204, 109832. https://doi.org/10.1016/j.envint.2025.109832
[2] Chen Y, Luo L, Hu S, Gan R, & Zeng L. (2023). The chemistry, processing, and preclinical anti-hyperuricemia potential of tea: a comprehensive review. Crit Rev Food Sci Nutr, 63(24), 7065-7090. https://doi.org/10.1080/10408398.2022.2040417
[3] Zhang Y, Li F, Liu L, An Z, Luo H, Zhang H, et al. (2025). Nanoparticle Drug Delivery Systems for Atherosclerosis: Precision Targeting, Inflammatory Modulation, and Plaque Stabilization. Adv Sci (Weinh), 12(36), e04990. https://doi.org/10.1002/advs.202504990
[4] Criqui MH, & Aboyans V. (2015). Epidemiology of peripheral artery disease. Circ Res, 116(9), 1509-1526. https://doi.org/10.1161/circresaha.116.303849
[5] Shubietah A, Awashra A, Milhem F, Ghannam M, Hattab M, Rajab I, et al. (2025). Hyperuricemia and Cardiovascular Risk: Insights and Implications. Crit Pathw Cardiol, 24(3), e0388. https://doi.org/10.1097/hpc.0000000000000388
[6] He B, Nie Q, Wang F, Wang X, Zhou Y, Wang C, et al. (2023). Hyperuricemia promotes the progression of atherosclerosis by activating endothelial cell pyroptosis via the ROS/NLRP3 pathway. J Cell Physiol, 238(8), 1808-1822. https://doi.org/10.1002/jcp.31038
[7] Burgess S, & Thompson SG. (2015). Multivariable Mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am J Epidemiol, 181(4), 251-260. https://doi.org/10.1093/aje/kwu283
[8] Sakaue S, Kanai M, Tanigawa Y, Karjalainen J, Kurki M, Koshiba S, et al. (2021). A cross-population atlas of genetic associations for 220 human phenotypes. Nat Genet, 53(10), 1415-1424. https://doi.org/10.1038/s41588-021-00931-x
[9] VanderWeele TJ, Tchetgen Tchetgen EJ, Cornelis M, & Kraft P. (2014). Methodological challenges in mendelian randomization. Epidemiology, 25(3), 427-435. https://doi.org/10.1097/ede.0000000000000081
[10] Palmer TM, Lawlor DA, Harbord RM, Sheehan NA, Tobias JH, Timpson NJ, et al. (2012). Using multiple genetic variants as instrumental variables for modifiable risk factors. Stat Methods Med Res, 21(3), 223-242. https://doi.org/10.1177/0962280210394459
[11] Polito L, Bortolotti M, Battelli MG, & Bolognesi A. (2022). Chronic kidney disease: Which role for xanthine oxidoreductase activity and products? Pharmacol Res, 184, 106407. https://doi.org/10.1016/j.phrs.2022.106407
[12] Burlina S, Dalfrà MG, Chilelli NC, & Lapolla A. (2016). Gestational Diabetes Mellitus and Future Cardiovascular Risk: An Update. Int J Endocrinol, 2016, 2070926. https://doi.org/10.1155/2016/2070926
[13] Zhou Y, Chen M, Zheng J, Shui X, He Y, Luo H, et al. (2024). Insights into the relationship between serum uric acid and pulmonary hypertension (Review). Mol Med Rep, 29(1). https://doi.org/10.3892/mmr.2023.13133
[14] Chen Q, Wang Z, Zhou J, Chen Z, Li Y, Li S, et al. (2020). Effect of Urate-Lowering Therapy on Cardiovascular and Kidney Outcomes: A Systematic Review and Meta-Analysis. Clin J Am Soc Nephrol, 15(11), 1576-1586. https://doi.org/10.2215/cjn.05190420
[15] Mirbolouk F, Arami S, Gholipour M, Khalili Y, Modallalkar SS, & Naghshbandi M. (2021). Is there any association between contrast-induced nephropathy and serum uric acid levels? J Cardiovasc Thorac Res, 13(1), 61-67. https://doi.org/10.34172/jcvtr.2021.20
[16] Zheng J, Yi Y, Tian T, Luo S, Liang X, & Bai Y. (2025). ICI-induced cardiovascular toxicity: mechanisms and immune reprogramming therapeutic strategies. Front Immunol, 16, 1550400. https://doi.org/10.3389/fimmu.2025.1550400
[17] Liu S, Zhong Z, & Liu F. (2022). Prognostic value of hyperuricemia for patients with sepsis in the intensive care unit. Sci Rep, 12(1), 1070. https://doi.org/10.1038/s41598-022-04862-3
[18] Li Y, Arai S, Kato K, Iwabuchi S, Iwabuchi N, Muto N, et al. (2023). The Potential Immunomodulatory Effect of Bifidobacterium longum subsp. longum BB536 on Healthy Adults through Plasmacytoid Dendritic Cell Activation in the Peripheral Blood. Nutrients, 16(1). https://doi.org/10.3390/nu16010042
[19] Nitz K, Herrmann J, Lerman A, & Lutgens E. (2024). Costimulatory and Coinhibitory Immune Checkpoints in Atherosclerosis: Therapeutic Targets in Atherosclerosis? JACC Basic Transl Sci, 9(6), 827-843. https://doi.org/10.1016/j.jacbts.2023.12.007
[20] Tan H, Zhang S, Zhang Z, Zhang J, Wang Z, Liao J, et al. (2024). Neutrophil extracellular traps promote M1 macrophage polarization in gouty inflammation via targeting hexokinase-2. Free Radic Biol Med, 224, 540-553. https://doi.org/10.1016/j.freeradbiomed.2024.09.009
[21] Kienhorst L, Janssens H, Radstake T, van Riel P, Jacobs J, van Koolwijk E, et al. (2017). A pilot study of CXCL8 levels in crystal proven gout patients during allopurinol treatment and their association with cardiovascular disease. Joint Bone Spine, 84(6), 709-713. https://doi.org/10.1016/j.jbspin.2016.10.013
[22] Thayaparan D, Emoto T, Khan AB, Besla R, Hamidzada H, El-Maklizi M, et al. (2025). Endothelial dysfunction drives atherosclerotic plaque macrophage-dependent abdominal aortic aneurysm formation. Nat Immunol, 26(5), 706-721. https://doi.org/10.1038/s41590-025-02132-8
[23] Charo IF, & Peters W. (2003). Chemokine receptor 2 (CCR2) in atherosclerosis, infectious diseases, and regulation of T-cell polarization. Microcirculation, 10(3-4), 259-264. https://doi.org/10.1038/sj.mn.7800191
Type
Published
Data Availability Statement
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
Issue
Section
License
Copyright (c) 2026 Cell Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.



