Puerarin Inhibits Gastric Cancer Cell Proliferationby Blocking the G2/M Phase Transition
DOI:
https://doi.org/10.71321/zf32x172Keywords:
Puerarin, Gastric cancer, Cell cycleAbstract
Background: Gastric cancer is a tumor with high morbidity and mortality with in the world, and according to the statistics of the World Health Organization (WHO), the incidence of gastric cancer is predominant in Asia, especially in East Asia, China and Japan are high-incidence areas, which is related to the special dietary habits of East Asians.
Methods: In this study, we first investigated the effects of puerarin on the biological behaviors of two gastric cancer cell lines, HGC27 and AGS. To further explore the underlying mechanisms, we performed transcriptome sequencing, which revealed that puerarin primarily influences gastric cancer progression by regulating cell cycle transitions. To validate these findings, we examined the expression levels of cell cycle-related proteins and analyzed cell cycle distribution using flow cytometry.
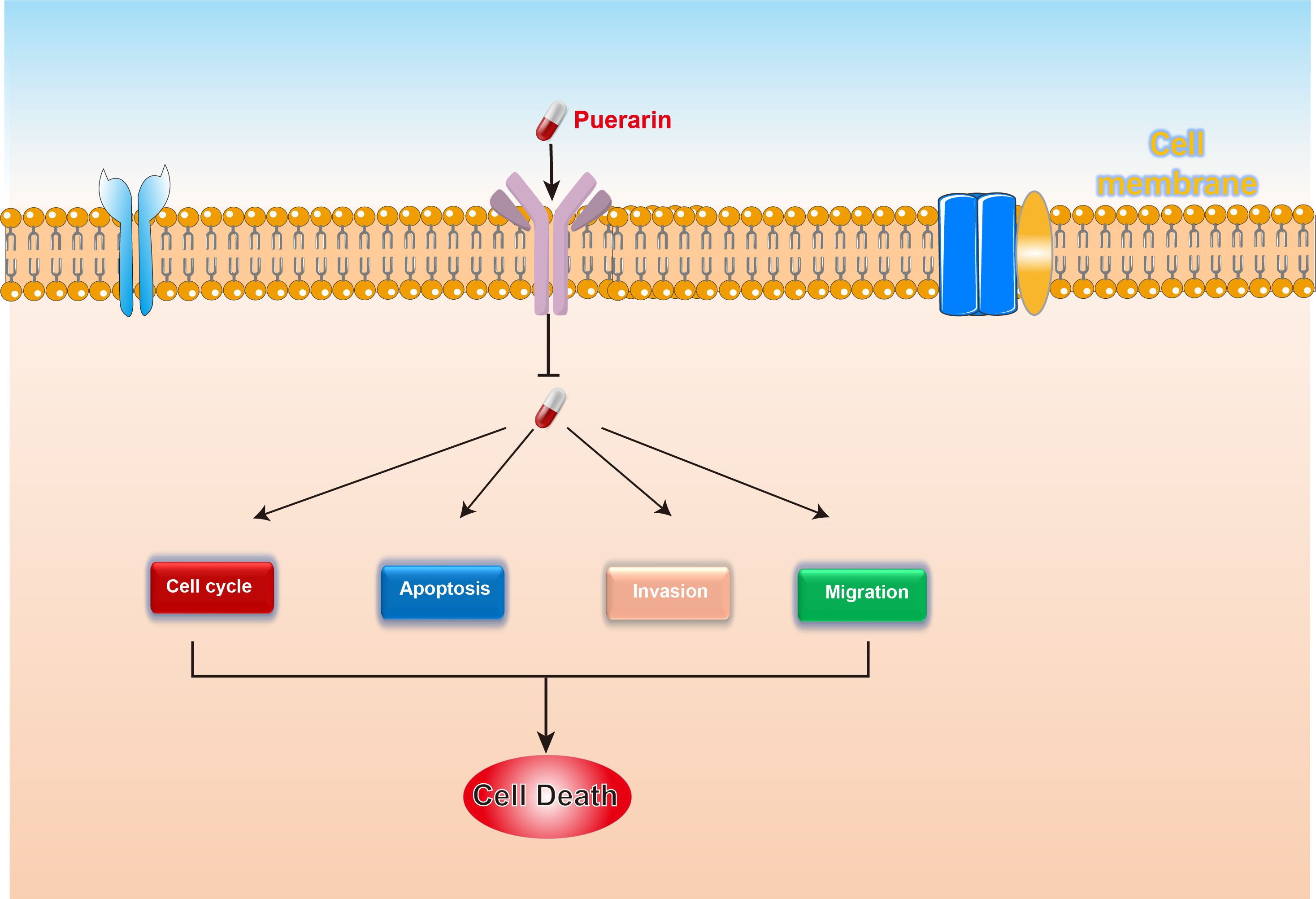
Results: This study demonstrated that puerarin significantly inhibits the proliferation, migration, and invasion of gastric cancer cells (HGC27 and AGS) while promoting their apoptosis. Transcriptome sequencing analysis revealed that puerarin primarily affects the biological behaviors of gastric cancer cells by regulating the G2-to-M phase transition. To validate this mechanism, we further employed flow cytometry to assess cell cycle distribution and analyzed the expression levels of cell cycle-related proteins, providing protein-level evidence that supports the G2/M phase transition regulation identified in the transcriptomic data.
Conclusion: Puerarin effectively inhibits the proliferation and invasion of gastric cancer cells. Its mechanism is closely related to the regulation of the G2-to-M phase transition, thereby affecting cell cycle progression and proliferative capacity.
References
[1] Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, & Et. A. (2020). Gastric cancer. The Lancet. https://doi.org/10.1016/S0140-6736(20)31288-5
[2] Chia N-Y, & Tan P. (2016). Molecular classification of gastric cancer. Annals of Oncology. https://doi.org/10.1093/annonc/mdw040
[3] Sexton RE, Al Hallak MN, Diab M, & Azmi AS. (2020). Gastric Cancer: A Comprehensive Review of Current and Future Treatment Strategies. Cancer Metastasis Rev, 39(4), 1179–1203. https://doi.org/10.1007/s10555-020-09925-3
[4] Rha SY, Oh D-Y, Yañez P, Bai Y, Ryu M-H, Lee J, et al. (2023). Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol, 24(11), 1181–1195. https://doi.org/10.1016/S1470-2045(23)00515-6
[5] Zheng X, Wang R, Zhang X, Sun Y, Zhang H, Zhao Z, et al. (2022). A deep learning model and human-machine fusion for prediction of EBV-associated gastric cancer from histopathology. Nat Commun, 13, 2790. https://doi.org/10.1038/s41467-022-30459-5
[6] Li W-Q, Zhang J-Y, Ma J-L, Li Z-X, Zhang L, Zhang Y, et al. (2019). Effects of Helicobacter pylori treatment and vitamin and garlic supplementation on gastric cancer incidence and mortality: follow-up of a randomized intervention trial. BMJ, 366, l5016. https://doi.org/10.1136/bmj.l5016
[7] Zeng Y, & Jin RU. (2022). Molecular pathogenesis, targeted therapies, and future perspectives for gastric cancer. Seminars in Cancer Biology, 86, 566–582. https://doi.org/10.1016/j.semcancer.2021.12.004
[8] Ma S, Zhou M, Xu Y, Gu X, Zou M, Abudushalamu G, et al. (2023). Clinical application and detection techniques of liquid biopsy in gastric cancer. Mol Cancer, 22, 7. https://doi.org/10.1186/s12943-023-01715-z
[9] Wang X, Zhang J, Cao G, Hua J, Shan G, & Lin W. (2022). Emerging roles of circular RNAs in gastric cancer metastasis and drug resistance. J Exp Clin Cancer Res, 41, 218. https://doi.org/10.1186/s13046-022-02432-z
[10] Suski JM, Braun M, Strmiska V, & Sicinski P. (2021). Targeting Cell-cycle Machinery in Cancer. Cancer Cell, 39(6), 759–778. https://doi.org/10.1016/j.ccell.2021.03.010
[11] Liu J, Peng Y, & Wei W. (2022). Cell cycle on the crossroad of tumorigenesis and cancer therapy. Trends Cell Biol, 32(1), 30–44. https://doi.org/10.1016/j.tcb.2021.07.001
[12] Evan GI, & Vousden KH. (2001). Proliferation, cell cycle and apoptosis in cancer. Nature, 411(6835), 342–348. https://doi.org/10.1038/35077213
[13] Engeland K. (2022). Cell cycle regulation: p53-p21-RB signaling. Cell Death Differ, 29(5), 946–960. https://doi.org/10.1038/s41418-022-00988-z
[14] Wang H, Guo M, Wei H, & Chen Y. (2023). Targeting p53 pathways: mechanisms, structures and advances in therapy. Signal Transduct Target Ther, 8, 92. https://doi.org/10.1038/s41392-023-01347-1
[15] Engeland K. (2018). Cell cycle arrest through indirect transcriptional repression by p53: I have a DREAM. Cell Death Differ, 25(1), 114–132. https://doi.org/10.1038/cdd.2017.172
[16] Bertoli C, Skotheim JM, & de Bruin RAM. (2013). Control of cell cycle transcription during G1 and S phases. Nat Rev Mol Cell Biol, 14(8), 518–528. https://doi.org/10.1038/nrm3629
[17] Icard P, Fournel L, Wu Z, Alifano M, & Lincet H. (2019). Interconnection between Metabolism and Cell Cycle in Cancer. Trends Biochem Sci, 44(6), 490–501. https://doi.org/10.1016/j.tibs.2018.12.007
[18] Targeting Cell-cycle Machinery in Cancer - PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC8206013/. Accessed 19 Jan 2026
[19] Zhou Y-X, Zhang H, & Peng C. (2013). Puerarin: A Review of Pharmacological Effects. Phytotherapy Research. https://doi.org/10.1002/ptr.5083
[20] Liu X, Huang R, & Wan J. (2023). Puerarin: a potential natural neuroprotective agent for neurological disorders. Biomedicine & Pharmacotherapy, 162, 114581. https://doi.org/10.1016/j.biopha.2023.114581
[21] Meng F, Guo B, Ma Y, Li K, & Niu F. (2022). Puerarin: A review of its mechanisms of action and clinical studies in ophthalmology. Phytomedicine, 107, 154465. https://doi.org/10.1016/j.phymed.2022.154465
[22] Lv J, Shi S, Zhang B, Xu X, Zheng H, Li Y, et al. (2022). Role of puerarin in pathological cardiac remodeling: A review. Pharmacological Research, 178, 106152. https://doi.org/10.1016/j.phrs.2022.106152
[23] Ye Y, Gao Y, Fang Y, Xu L, & He F. (2022). Anticancer Effect of Puerarin on Ovarian Cancer Progression Contributes to the Tumor Suppressor Gene Expression and Gut Microbiota Modulation. J Immunol Res, 2022, 4472509. https://doi.org/10.1155/2022/4472509
[24] ZENG Y-P, YANG Z-R, GUO X-F, JUN W, & DONG W-G. (2014). Synergistic effect of puerarin and 5-fluorouracil on hepatocellular carcinoma. Oncol Lett, 8(6), 2436–2442. https://doi.org/10.3892/ol.2014.2534
[25] Lang J, Guo Z, Xing S, Sun J, Qiu B, Shu Y, et al. (2022). Inhibitory role of puerarin on the A549 lung cancer cell line. Transl Cancer Res, 11(11), 4117–4125. https://doi.org/10.21037/tcr-22-2246
[26] Wang H, Wang Z, Zhang Z, Liu J, & Hong L. (2023). β-Sitosterol as a Promising Anticancer Agent for Chemoprevention and Chemotherapy: Mechanisms of Action and Future Prospects. Adv Nutr, 14(5), 1085–1110. https://doi.org/10.1016/j.advnut.2023.05.013
Type
Published
Data Availability Statement
Raw data have been deposited to National Center for Biotechnology Information (NCBI) under the BioProject number PRJNA1248124.
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







