The Multidimensional Regulatory Network of Osteoblast Differentiation and Targeted Therapeutic Strategies for OP
DOI:
https://doi.org/10.71321/rqm4ns12Keywords:
OP, osteoblast, differentiation, signaling, therapeutic paradigmAbstract
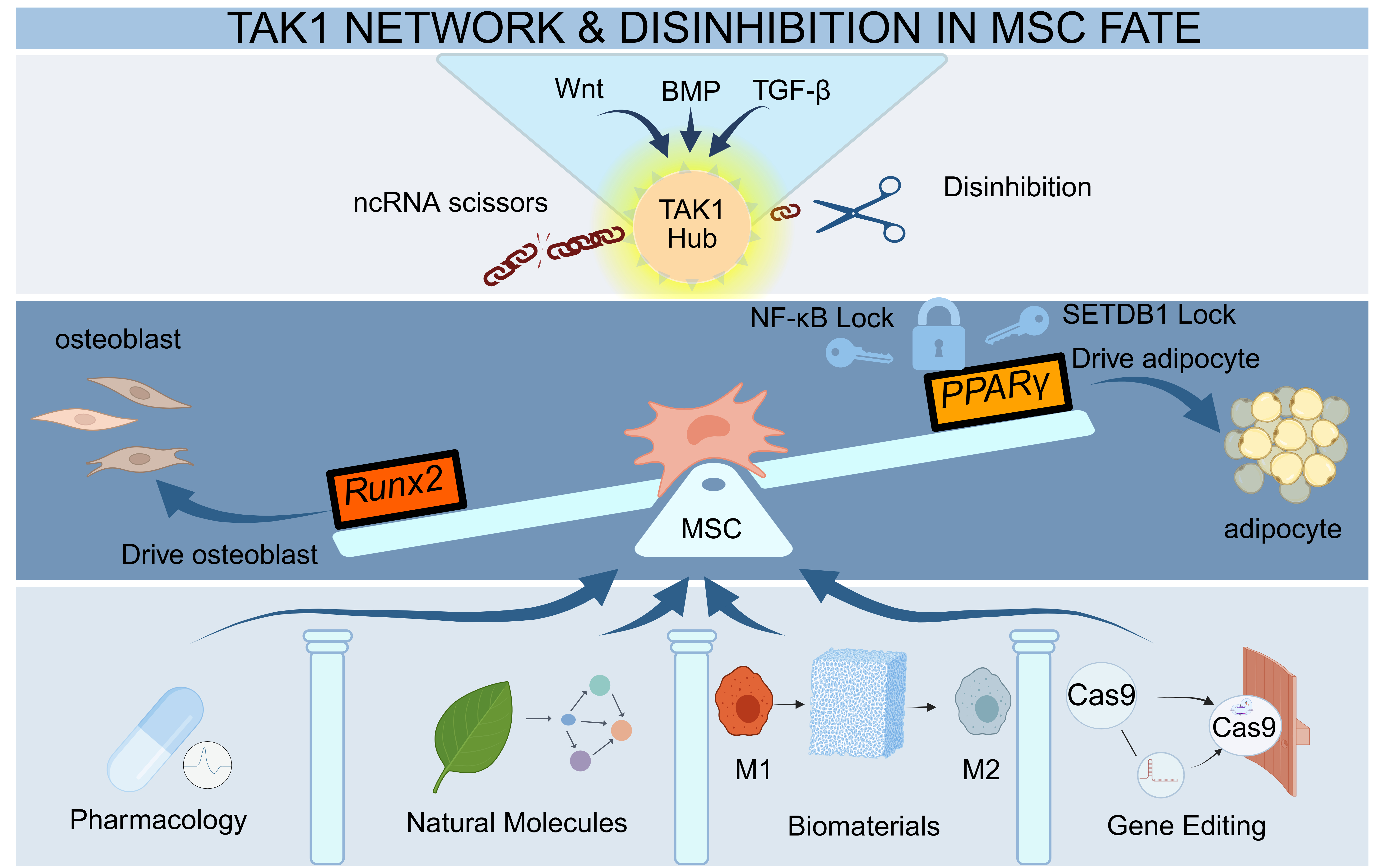
Osteoporosis (OP) represents a systemic failure of bone homeostasis, necessitating a therapeutic paradigm shift from broad-spectrum anti-resorptive agents to precision osteo-anabolic strategies. This review analyzes the multidimensional regulatory network governing osteoblast differentiation, emphasizing the transition from linear pathway descriptions to systems biology. We first elucidate the hierarchical integration of core signaling cascades—specifically the "context-dependent" crosstalk among Wnt, BMP, and TGF-β pathways—and their convergence on the master transcriptional hub, Runx2. A critical focus is placed on the pathological collapse of the Runx2-PPARγ equilibrium, which drives the inverse trajectory of osteogenic attenuation and marrow adiposity. Furthermore, we dissect the post-transcriptional "disinhibition" logic mediated by non-coding RNAs as a pivotal epigenetic layer. Bridging mechanistic insights with clinical translation, we classify emerging targeted strategies into three precision dimensions: pathology-dependent pharmacological reprogramming, osteoimmunomodulatory biomaterials with spatiotemporal responsiveness, and "scarless" gene editing technologies. Finally, we critically address pivotal translational bottlenecks—specifically the delivery barriers across the mineralized matrix and off-target safety risks—proposing that future therapeutics must evolve toward the precision homeostatic remodeling of the skeletal microenvironment.
References
[1] Stevens JR, Miranda-Carboni GA, Singer MA, Brugger SM, Lyons KM, Lane TF. Wnt10b deficiency results in age-dependent loss of bone mass and progressive reduction of mesenchymal progenitor cells. (2010). Journal of Bone and Mineral Research. 25(10) :2138–47. https://doi.org/10.1002/jbmr.118.
[2] Wu Z, Li W, Jiang K, Lin Z, Qian C, Wu M, et al. Regulation of bone homeostasis: signaling pathways and therapeutic targets. (2024). Medcomm. 5(8) :e657. https://doi.org/10.1002/mco2.657.
[3] Wu M, Wu S, Chen W, Li YP. The roles and regulatory mechanisms of TGF-β and BMP signaling in bone and cartilage development, homeostasis and disease. (2024). Cell Research. 34(2) :101–23. https://doi.org/10.1038/s41422-023-00918-9.
[4] Mikels AJ, Nusse R. Purified Wnt5a protein activates or inhibits β-catenin–TCF signaling depending on receptor context. (2006). PLOS Biology. 4(4) :e115. https://doi.org/10.1371/journal.pbio.0040115.
[5] Wu M, Chen G, Li YP. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. (2016). Bone Research. 4 :16009. https://doi.org/10.1038/boneres.2016.9.
[6] Włodarski KH. Properties and origin of osteoblasts. (1990). Clinical Orthopaedics and Related Research. (252) :276–93.
[7] Rahman MS, Akhtar N, Jamil HM, Banik RS, Asaduzzaman SM. TGF-β/BMP signaling and other molecular events: regulation of osteoblastogenesis and bone formation. (2015). Bone Research. 3 :15005. https://doi.org/10.1038/boneres.2015.5.
[8] Bonewald LF, Johnson ML. Osteocytes, mechanosensing and wnt signaling. (2008). Bone. 42(4) :606–15. https://doi.org/10.1016/j.bone.2007.12.224.
[9] Lin X, Patil S, Gao YG, Qian A. The bone extracellular matrix in bone formation and regeneration. (2020). Frontiers in Pharmacology. 11 :757. https://doi.org/10.3389/fphar.2020.00757.
[10] Reis TG, Del Colletto AMS, Silva LAS, Koga BAA, Sogayar MC, Carreira ACO. Recombinant human peptide growth factors, bone morphogenetic protein-7 (rhBMP7), and platelet-derived growth factor-BB (rhPDGF-BB) for osteoporosis treatment in an oophorectomized rat model. (2024). Biomolecules. 14(3) :317. https://doi.org/10.3390/biom14030317.
[11] Maeda K, Kobayashi Y, Udagawa N, Uehara S, Ishihara A, Mizoguchi T, et al. Wnt5a-Ror2 signaling between osteoblast-lineage cells and osteoclast precursors enhances osteoclastogenesis. (2012). Nature Medicine. 18(3) :405–12. https://doi.org/10.1038/nm.2653.
[12] Bolzoni M, Donofrio G, Storti P, Guasco D, Toscani D, Lazzaretti M, et al. Myeloma cells inhibit non-canonical wnt co-receptor ror2 expression in human bone marrow osteoprogenitor cells: effect of wnt5a/ror2 pathway activation on the osteogenic differentiation impairment induced by myeloma cells. (2013). Leukemia. 27(2) :451–63. https://doi.org/10.1038/leu.2012.190.
[13] Shen X, Feng S, Chen S, Gong B, Wang S, Wang H, et al. Wnt3a-induced LRP6 phosphorylation enhances osteoblast differentiation to alleviate osteoporosis through activation of mTORC1/β-catenin signaling. (2024). Archives of Biochemistry and Biophysics. 761 :110169. https://doi.org/10.1016/j.abb.2024.110169.
[14] Tamai K, Semenov M, Kato Y, Spokony R, Liu C, Katsuyama Y, et al. LDL-receptor-related proteins in wnt signal transduction. (2000). Nature. 407(6803) :530–5. https://doi.org/10.1038/35035117.
[15] He X, Semenov M, Tamai K, Zeng X. LDL receptor-related proteins 5 and 6 in wnt/beta-catenin signaling: arrows point the way. (2004). Development (cambridge, England). 131(8) :1663–77. https://doi.org/10.1242/dev.01117.
[16] Veeman MT, Axelrod JD, Moon RT. A second canon. Functions and mechanisms of beta-catenin-independent wnt signaling. (2003). Developmental Cell. 5(3) :367–77. https://doi.org/10.1016/s1534-5807(03)00266-1.
[17] Volleman TNE, Schol J, Morita K, Sakai D, Watanabe M. Wnt3a and wnt5a as potential chondrogenic stimulators for nucleus pulposus cell induction: a comprehensive review. (2020). Neurospine. 17(1) :19–35. https://doi.org/10.14245/ns.2040040.020.
[18] Jullien N, Maudinet A, Leloutre B, Ringe J, Häupl T, Marie PJ. Downregulation of ErbB3 by Wnt3a contributes to wnt-induced osteoblast differentiation in mesenchymal cells. (2012). Journal of Cellular Biochemistry. 113(6) :2047–56. https://doi.org/10.1002/jcb.24076.
[19] Huang K, Cai S, Fu T, Zhu Q, Liu L, Yao Z, et al. Wnt10b regulates osteogenesis of adipose-derived stem cells through wnt/β-catenin signalling pathway in osteoporosis. (2024). Cell Proliferation. 57(1) :e13522. https://doi.org/10.1111/cpr.13522.
[20] Liu Y, Fang J, Zhang Q, Zhang X, Cao Y, Chen W, et al. Wnt10b-overexpressing umbilical cord mesenchymal stem cells promote critical size rat calvarial defect healing by enhanced osteogenesis and VEGF-mediated angiogenesis. (2020). Journal of Orthopaedic Translation. 23 :29–37. https://doi.org/10.1016/j.jot.2020.02.009.
[21] Takada I, Suzawa M, Matsumoto K, Kato S. Suppression of PPAR transactivation switches cell fate of bone marrow stem cells from adipocytes into osteoblasts. (2007). Annals of the New York Academy of Sciences. 1116 :182–95. https://doi.org/10.1196/annals.1402.034.
[22] van Amerongen R, Fuerer C, Mizutani M, Nusse R. Wnt5a can both activate and repress wnt/β-catenin signaling during mouse embryonic development. (2012). Developmental Biology. 369(1) :101–14. https://doi.org/10.1016/j.ydbio.2012.06.020.
[23] Yang Y, Topol L, Lee H, Wu J. Wnt5a and Wnt5b exhibit distinct activities in coordinating chondrocyte proliferation and differentiation. (2003). Development (cambridge, England). 130(5) :1003–15. https://doi.org/10.1242/dev.00324.
[24] Chen G, Deng C, Li YP. TGF-β and BMP signaling in osteoblast differentiation and bone formation. (2012). International Journal of Biological Sciences. 8(2) :272–88. https://doi.org/10.7150/ijbs.2929.
[25] Guicheux J, Lemonnier J, Ghayor C, Suzuki A, Palmer G, Caverzasio J. Activation of p38 mitogen-activated protein kinase and c-jun-NH2-terminal kinase by BMP-2 and their implication in the stimulation of osteoblastic cell differentiation. (2003). Journal of Bone and Mineral Research: the Official Journal of the American Society for Bone and Mineral Research. 18(11) :2060–8. https://doi.org/10.1359/jbmr.2003.18.11.2060.
[26] Hunziker EB, Liu Y, Muff M, Haegi T, Shintani N, Lippuner K. The slow release of BMP-7 at a low dose accelerates dental implant healing in an osteopenic environment. (2021). European Cells and Materials. 41 :170–83. https://doi.org/10.22203/eCM.v041a12.
[27] Kim JA, Im S, Lim J, Hong JM, Ihn HJ, Bae JS, et al. The guanine nucleotide exchange factor DOCK5 negatively regulates osteoblast differentiation and BMP2-induced bone regeneration via the MKK3/6 and p38 signaling pathways. (2025). Experimental & Molecular Medicine. https://doi.org/10.1038/s12276-024-01372-2.
[28] Li ZZ, Wang HT, Lee GY, Yang Y, Zou YP, Wang B, et al. Bleomycin: a novel osteogenesis inhibitor of dental follicle cells via a TGF-β1/SMAD7/RUNX2 pathway. (2021). British Journal of Pharmacology. 178(2) :312–27. https://doi.org/10.1111/bph.15281.
[29] Almuraikhi N. Inhibition of TGF-β type I receptor by SB505124 down-regulates osteoblast differentiation and mineralization of human mesenchymal stem cells. (2023). Cell Biochemistry and Function. 41(5) :564–72. https://doi.org/10.1002/cbf.3812.
[30] Cao Z, Liu G, Zhang H, Wang M, Xu Y. Nox4 promotes osteoblast differentiation through TGF-beta signal pathway. (2022). Free Radical Biology & Medicine. 193(Pt 2) :595–609. https://doi.org/10.1016/j.freeradbiomed.2022.11.016.
[31] Liu DD, Zhang CY, Liu Y, Li J, Wang YX, Zheng SG. RUNX2 regulates osteoblast differentiation via the BMP4 signaling pathway. (2022). Journal of Dental Research. 101(10) :1227–37. https://doi.org/10.1177/00220345221093518.
[32] Komori T. Whole aspect of Runx2 functions in skeletal development. (2022). International journal of molecular sciences. 23(10) :5776. https://doi.org/10.3390/ijms23105776.
[33] Amir M, Jeevithan L, Barkat M, Fatima SH, Khan M, Israr S, et al. Advances in regenerative dentistry: a systematic review of harnessing wnt/β-catenin in dentin-pulp regeneration. (2024). Cells. 13(13) :1153. https://doi.org/10.3390/cells13131153.
[34] Yu S, Guo J, Sun Z, Lin C, Tao H, Zhang Q, et al. BMP2-dependent gene regulatory network analysis reveals Klf4 as a novel transcription factor of osteoblast differentiation. (2021). Cell Death and Disease. 12(2) :197. https://doi.org/10.1038/s41419-021-03480-7.
[35] Piao M, Lee SH, Kim MJ, Choi JK, Yeo CY, Lee KY. Cyclophilin a promotes osteoblast differentiation by regulating Runx2. (2022). International journal of molecular sciences. 23(16) :9244. https://doi.org/10.3390/ijms23169244.
[36] Liu M, Goldman G, MacDougall M, Chen S. BMP signaling pathway in dentin development and diseases. (2022). Cells. 11(14) :2216. https://doi.org/10.3390/cells11142216.
[37] Chiba N, Noguchi Y, Seong CH, Ohnishi T, Matsuguchi T. EGR1 plays an important role in BMP9-mediated osteoblast differentiation by promoting SMAD1/5 phosphorylation. (2022). FEBS Letters. 596(13) :1720–32. https://doi.org/10.1002/1873-3468.14407.
[38] Dashti P, Thaler R, Hawse JR, Galvan ML, van der Eerden BJ, van Wijnen AJ, et al. G-protein coupled receptor 5C (GPRC5C) is required for osteoblast differentiation and responds to EZH2 inhibition and multiple osteogenic signals. (2023). Bone. 176 :116866. https://doi.org/10.1016/j.bone.2023.116866.
[39] Zhou J, Yang J, Dong Y, Shi Y, Zhu E, Yuan H, et al. Oncostatin M receptor regulates osteoblast differentiation via extracellular signal-regulated kinase/autophagy signaling. (2022). Stem Cell Research & Therapy. 13(1) :278. https://doi.org/10.1186/s13287-022-02958-1.
[40] Zhou Q, Zhou L, Li J. MiR-218-5p-dependent SOCS3 downregulation increases osteoblast differentiation inpostmenopausal osteoporosis. (2023). Journal of Orthopaedic Surgery and Research. 18(1) :109. https://doi.org/10.1186/s13018-023-03580-4.
[41] Zhao H, Yang Y, Wang Y, Feng X, Deng A, Ou Z, et al. MicroRNA-497-5p stimulates osteoblast differentiation through HMGA2-mediated JNK signaling pathway. (2020). Journal of Orthopaedic Surgery and Research. 15(1) :515. https://doi.org/10.1186/s13018-020-02043-4.
[42] Hu L, Liu J, Xue H, Panayi AC, Xie X, Lin Z, et al. miRNA-92a-3p regulates osteoblast differentiation in patients with concomitant limb fractures and TBI via IBSP/PI3K-AKT inhibition. (2021). Molecular Therapy Nucleic Acids. 23 :1345–59. https://doi.org/10.1016/j.omtn.2021.02.008.
[43] Liu F, Liang Y, Lin X. MiR-151b inhibits osteoblast differentiation via downregulating Msx2. (2022). Connective Tissue Research. 63(2) :112–23. https://doi.org/10.1080/03008207.2021.1882442.
[44] Zhang L, Zeng C, Huang J, Yan H, Jiang Y, Li R. ExploratiomiR-n of the 187-3p/CNR2 pathway in modulating osteoblast differentiation and treating postmenopausal osteoporosis through mechanical stress. (2024). FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 38(13) :e23776. https://doi.org/10.1096/fj.202400113RR.
[45] Zhai S, Liu C, Vimalraj S, Subramanian R, Abullais SS, Arora S, et al. Glucagon-like peptide-1 receptor promotes osteoblast differentiation of dental pulp stem cells and bone formation in a zebrafish scale regeneration model. (2023). Peptides. 163 :170974. https://doi.org/10.1016/j.peptides.2023.170974.
[46] Tan AQ, Zheng YF. The roles of SNHG family in osteoblast differentiation. (2022). Genes. 13(12) :2268. https://doi.org/10.3390/genes13122268.
[47] Chen L, Liu H, Sun C, Pei J, Li J, Li Y, et al. A novel LncRNA SNHG3 promotes osteoblast differentiation through BMP2 upregulation in aortic valve calcification. (2022). JACC Basic to translational science. 7(9) :899–914. https://doi.org/10.1016/j.jacbts.2022.06.009.
[48] Wang Z, Zhou Y, Dai Z, Chen X, Li C, Lin Z, et al. A novel long noncoding RNA, lnc-OAD, is required for bone morphogenetic protein 2- (BMP-2-) induced osteoblast differentiation. (2021). Biomed Research International. 2021 :6697749. https://doi.org/10.1155/2021/6697749.
[49] Morita M, Arshad F, Quayle LA, George CN, Lefley DV, Kalajzic I, et al. Losartan alters osteoblast differentiation and increases bone mass through inhibition of TGFB signalling in vitro and in an OIM mouse model. (2024). Bone Reports. 22 :101795. https://doi.org/10.1016/j.bonr.2024.101795.
[50] Muthurangan M, Elsafadi M, Siyal A, Kaimkhani ZA, Umrani A, AlMuraikhi NA, et al. Transient downregulation of NR4A1 leads to impaired osteoblast differentiation through the TGF-ß pathway, and elesclomol (STA-4783) rescues this phenotype. (2023). Cell Biochemistry and Function. 41(5) :590–8. https://doi.org/10.1002/cbf.3814.
[51] Chen JH, Shen C, Oh H, Park JH. Exendin-4, a glucagon-like peptide receptor agonist, facilitates osteoblast differentiation via connexin43. (2021). Endocrine. 72(3) :672–80. https://doi.org/10.1007/s12020-021-02664-7.
[52] Lai YC, Yao ZK, Chang TC, Feng CW, Kuo TJ, Luo YW, et al. Dextromethorphan inhibits osteoblast differentiation and bone regeneration of rats with subcritical-sized calvarial defects. (2024). Environmental Toxicology. https://doi.org/10.1002/tox.24447.
[53] Tan L, Miao Z, Zhao Y, Liang Y, Xu N, Chen X, et al. Dual regulation of phaseol on osteoclast formation and osteoblast differentiation by targeting TAK1 kinase for osteoporosis treatment. (2024). Journal of Advanced Research. :S2090-1232(24)565-4. https://doi.org/10.1016/j.jare.2024.12.009.
[54] Constanze B, Popper B, Aggarwal BB, Shakibaei M. Evidence that TNF-β suppresses osteoblast differentiation of mesenchymal stem cells and resveratrol reverses it through modulation of NF-κB, Sirt1 and Runx2. (2020). Cell and Tissue Research. 381(1) :83–98. https://doi.org/10.1007/s00441-020-03188-8.
[55] Yun HM, Kim B, Jeong YH, Hong JT, Park KR. Suffruticosol a elevates osteoblast differentiation targeting BMP2-smad/1/5/8-RUNX2 in pre-osteoblasts. (2023). BioFactors (Oxford, England). 49(1) :127–39. https://doi.org/10.1002/biof.1878.
[56] Zhang T, Gao Y, Cui W, Li Y, Xiao D, Zhou R. Nanomaterials-based cell osteogenic differentiation and bone regeneration. (2021). Current Stem Cell Research & Therapy. 16(1) :36–47. https://doi.org/10.2174/1574888X15666200521083834.
[57] Gao K, Wang X, Wang Z, He L, Lin J, Bai Z, et al. Design of novel functionalized collagen-chitosan-MBG scaffolds for enhancing osteoblast differentiation in BMSCs. (2021). Biomedical Materials (Bristol, England). 16(6). https://doi.org/10.1088/1748-605X/ac3146.
[58] Chen K, Ha S, Xu L, Liu C, Liu Y, Wu X, et al. Fluorinated hydroxyapatite conditions a favorable osteo-immune microenvironment via triggering metabolic shift from glycolysis to oxidative phosphorylation. (2024). Journal of Translational Medicine. 22(1) :437. https://doi.org/10.1186/s12967-024-05261-0.
[59] Bone-targeted pH-responsive cerium nanoparticles for anabolic therapy in osteoporosis. (2021). Bioactive Materials. 6(12) :4697–706. https://doi.org/10.1016/j.bioactmat.2021.04.038.
[60] Huang XY, Zhou XX, Yang H, Xu T, Dao JW, Bian L, et al. Directed osteogenic differentiation of human bone marrow mesenchymal stem cells via sustained release of BMP4 from PBVHx-based nanoparticles. (2024). International Journal of Biological Macromolecules. 265 :130649. https://doi.org/10.1016/j.ijbiomac.2024.130649.
[61] Hinton PV, Genoud KJ, Early JO, O’Brien FJ, Kennedy OD. Impact of fluid flow shear stress on osteoblast differentiation and cross-talk with articular chondrocytes. (2022). International journal of molecular sciences. 23(16) :9505. https://doi.org/10.3390/ijms23169505.
[62] Hu J, Cui Z, Huang K, Su R, Zhao S. Role of the GRP78-c-src signaling pathway on osteoblast differentiation of periodontal ligament fibroblasts induced by cyclic mechanical stretch. (2024). Hua Xi Kou Qiang Yi Xue Za Zhi = Huaxi Kouqiang Yixue Zazhi = West China Journal of Stomatology. 42(3) :304–12. https://doi.org/10.7518/hxkq.2024.2023354.
[63] Chaugule S, Constantinou CK, John AA, Micha D, Eekhoff M, Gravallese E, et al. Comprehensive review of osteogenesis imperfecta: current treatments and future innovations. (2025). Human Gene Therapy. https://doi.org/10.1089/hum.2024.191.
[64] Morello R. Osteogenesis imperfecta and therapeutics. (2018). Matrix Biology. 71–72 :294–312. https://doi.org/10.1016/j.matbio.2018.03.010.
[65] Liang Y, Xu X, Xu L, Iqbal Z, Ouyang K, Zhang H, et al. Chondrocyte-specific genomic editing enabled by hybrid exosomes for osteoarthritis treatment. (2022). Theranostics. 12(11) :4866–78. https://doi.org/10.7150/thno.69368.
[66] Botor M, Fus-Kujawa A, Uroczynska M, Stepien KL, Galicka A, Gawron K, et al. Osteogenesis imperfecta: current and prospective therapies. (2021). Biomolecules. 11(10) :1493. https://doi.org/10.3390/biom11101493.
[67] Jiang Y, Zhang P, Zhang X, Lv L, Zhou Y. Advances in mesenchymal stem cell transplantation for the treatment of osteoporosis. https://doi.org/10.1111/cpr.12956.
[68] Han X, Alu A, Liu H, Shi Y, Wei X, Cai L. Biomaterial-assisted biotherapy: a brief review of biomaterials used in drug delivery, vaccine development, gene therapy, and stem cell therapy. (2022). Bioactive Materials. 17 :29–48. https://doi.org/10.1016/j.bioactmat.2022.01.011.
[69] Song S, Xia H, Guo M, Wang S, Zhang S, Ma P, et al. Role of macrophage in nanomedicine-based disease treatment. (2021). Drug Delivery.
[70] Ariffin SHZ, Lim KW, Wahab RMA, Ariffin ZZ, Din RDR, Shahidan MA, et al. Gene expression profiles for in vitro human stem cell differentiation into osteoblasts and osteoclasts: a systematic review. (2022). PeerJ. 10 :e14174. https://doi.org/10.7717/peerj.14174.
Type
Published
Data Availability Statement
None.
Issue
Section
License
Copyright (c) 2026 Cell Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.



