Advantages in Lung Cancer Diagnosis: a Recent Review
DOI:
https://doi.org/10.71321/cvmbxc42Keywords:
Lung cancer diagnosis, Imaging technologies, Biopsy, AI in radiology, biomarker discoveryAbstract
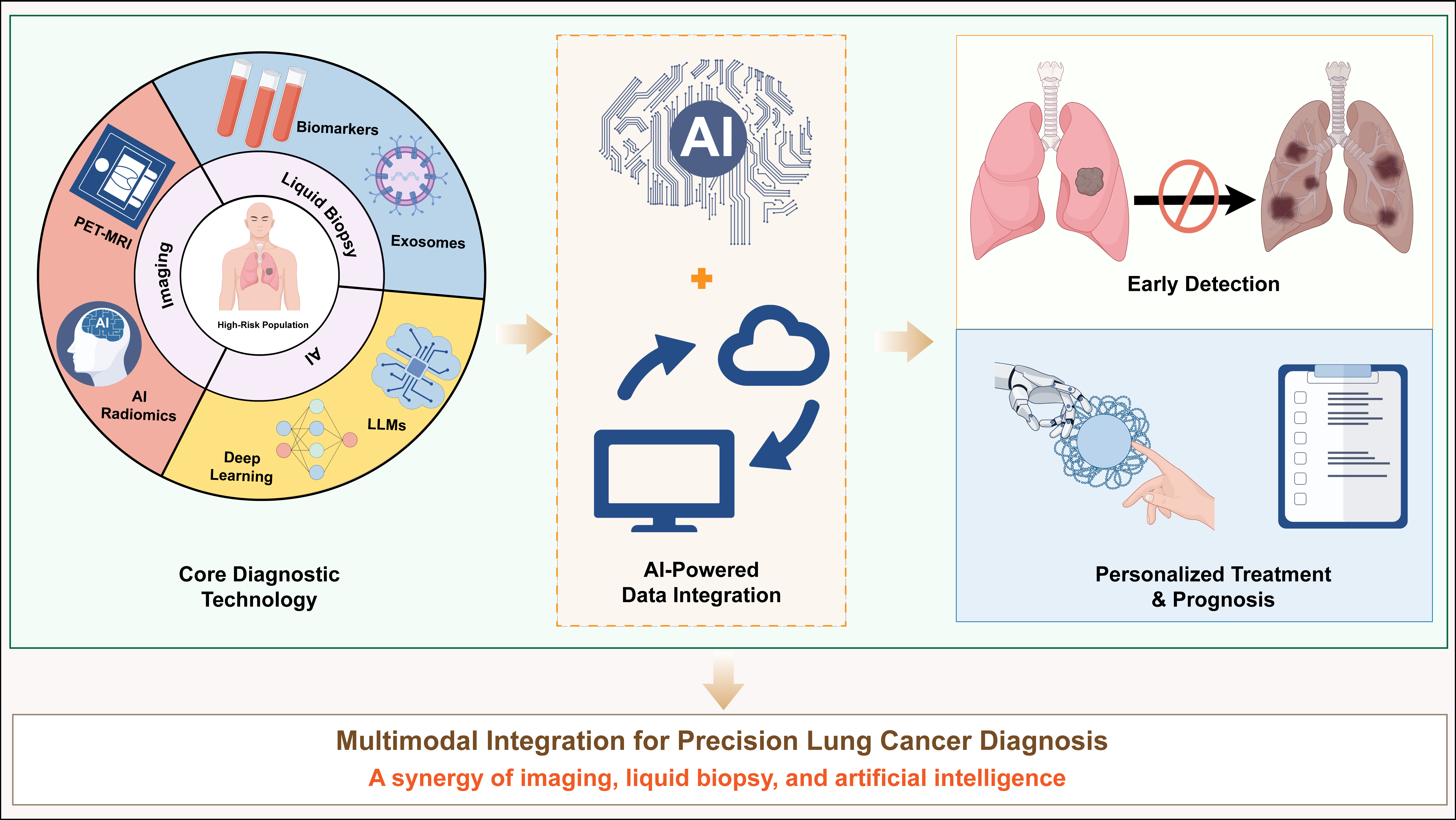
Lung cancer remains a leading cause of cancer-related mortality worldwide, underscoring the critical need for advancements in early and accurate diagnostic strategies. This review synthesizes recent progress in lung cancer diagnostics, focusing on innovations in imaging technologies, biomarker discovery, liquid biopsy, and artificial intelligence (AI)-driven analytical tools. Low-dose computed tomography (LDCT) has solidified its role in screening high-risk populations, reducing mortality through early detection, yet challenges persist regarding false positives and overdiagnosis. Emerging imaging modalities, including spectral CT and PET-MRI hybrid systems, demonstrate enhanced specificity in characterizing pulmonary nodules. AI and machine learning algorithms are increasingly deployed to refine radiological interpretation, predict malignancy risk, and correlate imaging features with molecular profiles. Despite these breakthroughs, challenges such as standardization, cost, and integration into clinical workflows remain. Future research would emphasize the synergy of multimodal diagnostics, leveraging AI to harmonize imaging, liquid biopsy, and clinical data for precision medicine.
References
[1] Bray, F., Laversanne, M., Sung, H., Ferlay, J., Siegel, R.L., Soerjomataram, I., et al. (2024). Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 74(3), 229-263. https://doi.org/10.3322/caac.21834.
[2] Hirsch, F.R., Scagliotti, G.V., Mulshine, J.L., Kwon, R., Curran, W.J., Wu, Y.-L., et al. (2017). Lung cancer: current therapies and new targeted treatments. Lancet, 389(10066), 299-311. https://doi.org/10.1016/S0140-6736(16)30958-8.
[3] Sung, H., Ferlay, J., Siegel, R.L., Laversanne, M., Soerjomataram, I., Jemal, A., et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 71(3), 209-249. https://doi.org/10.3322/caac.21660.
[4] Wang, J., Li, Z., Chen, Y., Luo, N., and He, S. (2024). Gelatin nanocarriers assembled by a self-immolative cross-linker for targeted cancer therapy. International journal of biological macromolecules, 268, 131722. https://doi.org/10.1016/j.ijbiomac.2024.131722.
[5] Khan, J.A., Albalkhi, I., Garatli, S., and Migliore, M. (2024). Recent advancements in minimally invasive surgery for early stage non-small cell lung cancer: a narrative review. Journal of clinical medicine, 13(11), 3354. https://doi.org/10.3390/jcm13113354.
[6] Madhani-Lovely, F., Graham, K., Graham, W., and Doheny, R. (2024). Late breaking abstract -reconstructing a lung cancer screening program to improve early stage lung cancer detection. European Respiratory Journal, 64, PA3343. https://doi.org/10.1183/13993003.congress-2024.PA3343.
[7] Sassorossi, C., Evangelista, J., Stefani, A., Chiappetta, M., Martino, A., Campanella, A., et al. (2025). The role of ctDNA for diagnosis and histological prediction in early stage non-small-cell lung cancer: a narrative review. Diagnostics, 15(7), 904. https://doi.org/10.3390/diagnostics15070904.
[8] de-Torres, J.P., Alcaide, A.B., Campo, A., Zulueta, J.J., Bastarrika, G., Ezponda, A., et al. (2024). Lung cancer screening in people with COPD: the pamplona-IELCAP experience. Archivos de bronconeumologia, 60(2), 95-100. https://doi.org/10.1016/j.arbres.2023.12.012.
[9] Grenier, P.A., Arutkin, M., Brun, A.L., Metivier, A.-C., Sage, E., Haziza, F., et al. (2024). Prevalent findings on low-dose CT scan lung cancer screening: a French prospective pilot study. European journal of public health, 35(2), 342-346. https://doi.org/10.1093/eurpub/ckae183.
[10] Reck, M., Dettmer, S., Kauczor, H.-U., Kaaks, R., Reinmuth, N., and Vogel-Claussen, J. (2023). Lung cancer screening with low-dose computed tomography. Deutsches Ärzteblatt International, 120(23), 387. https://doi.org/10.3238/arztebl.m2023.0099.
[11] Wu, J., Li, R., Zhang, H., Zheng, Q., Tao, W., Yang, M., et al. (2024). Screening for lung cancer using thin-slice low-dose computed tomography in southwestern China: a population-based real-world study. Thoracic cancer, 15(19), 1522-1532. https://doi.org/10.1111/1759-7714.15383.
[12] Beshiri, K., Marinello, A., Vasseur, D., Zullo, L., Sanchez Becerra, M.V., Pagliaro, A., et al. (2024). Clinical utility of liquid biopsy in early stage lung cancer: A single institution experience. Cancer Research, 84(6), 7490. https://doi.org/10.1158/1538-7445.am2024-7490.
[13] Gristina, V., La Mantia, M., Peri, M., Iacono, F., Barraco, N., Perez, A., et al. (2023). Navigating the liquid biopsy Minimal Residual Disease (MRD) in non-small cell lung cancer: Making the invisible visible. Critical reviews in oncology/hematology, 182, 103899. https://doi.org/10.1016/j.critrevonc.2022.103899.
[14] Kemper, M., Krekeler, C., Menck, K., Lenz, G., Evers, G., Schulze, A.B., et al. (2023). Liquid biopsies in lung cancer. Cancers, 15(5), 1430. https://doi.org/10.3390/cancers15051430.
[15] Vandekerckhove, O., Cuppens, K., Pat, K., Du Pont, B., Froyen, G., and Maes, B. (2023). Liquid biopsy in early-stage lung cancer: current and future clinical applications. Cancers, 15(10), 2702. https://doi.org/10.3390/cancers15102702.
[16] Grigoryeva, E.S., Tashireva, L.A., Savelieva, O.E., Zavyalova, M.V., Popova, N.O., Kuznetsov, G.A., et al. (2023). The association of integrins β3, β4, and αVβ5 on exosomes, CTCs and tumor cells with localization of distant metastasis in breast cancer patients. International journal of molecular sciences, 24(3), 2929. https://doi.org/10.3390/ijms24032929.
[17] Li, C., Shao, J., Li, P., Feng, J., Li, J., and Wang, C. (2023). Circulating tumor DNA as liquid biopsy in lung cancer: Biological characteristics and clinical integration. Cancer letters, 577, 216365. https://doi.org/10.1016/j.canlet.2023.216365.
[18] Papakonstantinou, D., Roumeliotou, A., Pantazaka, E., Shaukat, A.-N., Christopoulou, A., Koutras, A., et al. (2024). Integrative analysis of circulating tumor cells (CTCs) and exosomes from small-cell lung cancer (SCLC) patients: a comprehensive approach. Molecular oncology, DOI10.1002/1878-0261.13765. https://doi.org/10.1002/1878-0261.13765.
[19] She, W., Garitaonaindia, Y., and Lin, Y. (2024). The latest advances in liquid biopsy for lung cancer-a narrative review. Translational lung cancer research, 13(11), 3241-3251. https://doi.org/10.21037/tlcr-24-828.
[20] Hofman, P., Ourailidis, I., Romanovsky, E., Ilie, M., Budczies, J., and Stenzinger, A. (2025). Artificial intelligence for diagnosis and predictive biomarkers in Non-Small cell lung cancer Patients: New promises but also new hurdles for the pathologist. Lung Cancer, 200, 108110. https://doi.org/10.1016/j.lungcan.2025.108110.
[21] Upaisilsathaporn, P., Klawandee, S., and Wanchaijiraboon, P. (2024). The potential of chest X-ray artificial intelligence detecting missed lung cancer diagnosis in a community based cancer in thailand. Journal of Thoracic Oncology, 19(10), S473-S474.
[22] Yang, D., Miao, Y., Liu, C., Zhang, N., Zhang, D., Guo, Q., et al. (2024). Advances in artificial intelligence applications in the field of lung cancer. Frontiers in oncology, 14, 1449068. https://doi.org/10.3389/fonc.2024.1449068.
[23] Bao, J., Li, J., Zhao, L., and Cui, R. (2023). Diagnosis and assessment of lung cancer based on new imaging technologies. Asian journal of surgery, 46(7), 2853-2854. https://doi.org/10.1016/j.asjsur.2023.01.077.
[24] Nathani, A., and Dincer, H.E. (2025). Advancements in imaging technologies for the diagnosis of lung cancer and other pulmonary diseases. Diagnostics, 15(7), 826. https://doi.org/10.3390/diagnostics15070826.
[25] Guo, J., Liao, Z., and Dong, A. (2023). FDG PET/CT in a case of lung adenocarcinoma with diffuse cavitary intrapulmonary metastases. Clinical nuclear medicine, 48(5), 428-430. https://doi.org/10.1097/RLU.0000000000004534.
[26] Zhu, L., Liu, J., Zeng, L., Moonindranath, S., An, P., Chen, H., et al. (2024). Thoracic high resolution computed tomography evaluation of imaging abnormalities of 108 lung cancer patients with different pulmonary function. Cancer Imaging, 24(1), 78. https://doi.org/10.1186/s40644-024-00720-9.
[27] Liu, Y., Geng, Q., Lin, X., Feng, C., Qiao, Y., and Zhang, S. (2024). Benefits, harms, and cost-effectiveness of risk model-based and risk factor-based low-dose computed tomography screening strategies for lung cancer: a systematic review. BMC Cancer, 24(1), 1567. https://doi.org/10.1186/s12885-024-13356-6.
[28] Zhang, J., Shangguan, Z., Gong, W., and Cheng, Y. (2023). A novel denoising method for low-dose CT images based on transformer and CNN. Computers in biology and medicine, 163, 107162. https://doi.org/10.1016/j.compbiomed.2023.107162.
[29] Chen, L., Gu, D., Chen, Y., Shao, Y., Cao, X., Liu, G., et al. (2021). An artificial-intelligence lung imaging analysis system (ALIAS) for population-based nodule computing in CT scans. Computerized Medical Imaging and Graphics, 89, 101899. https://doi.org/10.1016/j.compmedimag.2021.101899.
[30] Liang, F., Li, C., and Fu, X. (2021). Evaluation of the effectiveness of artificial intelligence chest CT lung nodule detection based on deep learning. Journal of healthcare engineering, 2021, 9971325. https://doi.org/10.1155/2021/9971325.
[31] Pei, Q., Luo, Y., Chen, Y., Li, J., Xie, D., and Ye, T. (2022). Artificial intelligence in clinical applications for lung cancer: diagnosis, treatment and prognosis. Clinical chemistry and laboratory medicine, 60(12), 1974-1983. https://doi.org/10.1515/cclm-2022-0291.
[32] Yuan, L., An, L., Zhu, Y., Duan, C., Kong, W., Jiang, P., et al. (2024). Machine learning indiagnosis and prognosis of lung cancer by PET-CT. Cancer management and research, 16, 361-375. https://doi.org/10.2147/CMAR.S451871.
[33] Tarnoki, A.D., Tarnoki, D.L., Dabrowska, M., Knetki-Wroblewska, M., Frille, A., Stubbs, H., et al. (2024). New developments in the imaging of lung cancer. Breathe, 20(1), 230176-230176. https://doi.org/10.1183/20734735.0176-2023.
[34] Kausaite-Minkstimiene, A., Popov, A., Kalvaityte, U., Bernotiene, E., Mobasheri, A., and Ramanaviciene, A. (2023). An ultra-sensitive SPR immunosensor for quantitative determination of human cartilage oligomeric matrix protein biomarker. Biosensors & bioelectronics, 234, 115370. https://doi.org/10.1016/j.bios.2023.115370.
[35] Li, H., Huang, T., Yuan, H., Lu, L., Cao, Z., Zhang, L., et al. (2023). Combined ultrasensitive detection of renal cancer proteins and cells using an optical microfiber functionalized with Ti3C2 mXene and gold nanorod-nanosensitized interfaces. Analytical chemistry, 95(11), 5142-5150. https://doi.org/10.1021/acs.analchem.3c00281.
[36] Sun, J., Ning, X., Cui, L., Ling, M., Xu, X., and He, S. (2020). Assembly of "carrier free" enzymatic nano-reporters for improved ELISA. The Analyst, 145(20), 6541-6548. https://doi.org/10.1039/d0an00585a.
[37] Chu, J., Ejaz, A., Lin, K.M., Joseph, M.R., Coraor, A.E., Drummond, D.A., et al. (2024). Single-molecule fluorescence multiplexing by multi-parameter spectroscopic detection of nanostructured FRET labels. Nature nanotechnology, 19(8), 1150–1157. https://doi.org/10.1038/s41565-024-01672-8.
[38] Son, Y., Jeong, M., Lim, J.M., Venkatesh, K., Karuppiah, C., and Lee, H.J. (2025). Advances in spectroscopic detection of traumatic brain injury biomarkers: Potential for diagnostic applications. Applied Spectroscopy Reviews, 60, 889-918. https://doi.org/10.1080/05704928.2025.2483396.
[39] Gao, Z., Chen, Z., Deng, J., Li, X., Qu, Y., Xu, L., et al. (2018). Measurement of carcinoembryonic antigen in clinical serum samples using a centrifugal microfluidic device. Micromachines, 9(9), 470. https://doi.org/10.3390/mi9090470.
[40] Li, R., Fan, H., Zhou, H., Chen, Y., Yu, Q., Hu, W., et al. (2023). Nanozyme-catalyzed metasurface plasmon sensor-based portable ultrasensitive optical quantification platform for cancer biomarker screening. Advanced science (Weinheim, Baden-Wurttemberg, Germany), 10(24), e2301658. https://doi.org/10.1002/advs.202301658.
[41] Cao, Y., Chen, Y., Zhang, X., Zeng, H., Cui, L., and He, S. (2025). Biotinylation-based lateral flow assays for pathogenic and total bacteria detection. Analytica chimica acta, 1338, 343607. https://doi.org/10.1016/j.aca.2025.343607.
[42] He, S. (2023). No-cost ballpoint pen dispenser for lateral flow assays. Talanta, 263, 124742. https://doi.org/10.1016/j.talanta.2023.124742.
[43] Li, J., Cao, X., Zhang, L., Liu, A., Liu, S., Chen, F., et al. (2025). Anti-FDX1 autoantibody as a potential biomarker for non-small cell lung cancer detection. Cancer epidemiology, biomarkers & prevention, 34(3), 439-447. https://doi.org/10.1158/1055-9965.EPI-24-1096.
[44] Tobi, M., Ezekwudo, D., Tobi, Y.Y., Zhao, X., Antaki, F., Rambus, M., et al. (2025). Historic p87 Is diagnostic for lung cancer preceding clinical presentation by at least 4 years. Cancer, 17(6), 952. https://doi.org/10.3390/cancers17060952.
[45] Gidwani, K., Kekki, H., Terava, J., Soukka, T., Sundfeldt, K., and Pettersson, K. (2020). Nanoparticle-aided glycovariant assays to bridge biomarker performance and ctDNA results. Molecular aspects of medicine, 72, 100831. https://doi.org/10.1016/j.mam.2019.11.001.
[46] Pei, X., Wu, X., Xiong, J., Wang, G., Tao, G., Ma, Y., et al. (2020). Competitive aptasensor for the ultrasensitive multiplexed detection of cancer biomarkers by fluorescent nanoparticle counting. The Analyst, 145(10), 3612-3619. https://doi.org/10.1039/d0an00239a.
[47] Swami, S., Kayenat, F., and Wajid, S. (2024). SPR biosensing: Cancer diagnosis and biomarkers quantification. Microchemical Journal, 197, 109792. https://doi.org/10.1016/j.microc.2023.109792.
[48] Chen, Y., Chen, Y., Tang, S., Tang, B., and He, S. (2024). Real-time fluorescence growth curves for viable bacteria quantification in foods. Food chemistry: X, 24, 101886. https://doi.org/10.1016/j.fochx.2024.101886.
[49] Chen, Y., Sun, J., Luo, M., Cao, Y., Chen, Q., and He, S. (2024). Quantifying the viability of lactic acid bacteria using ratiometric fluorescence assays. Microchemical Journal, 206, 111485. https://doi.org/10.1016/j.microc.2024.111485.
[50] He, S., Chen, Y., Wang, J., Sun, J., Zhang, X., and Chen, Q. (2024). Rapid and sensitive quantification of bacterial viability using ratiometric fluorescence sensing. Analytical chemistry, 96(27), 11018-11025. https://doi.org/10.1021/acs.analchem.4c01737.
[51] Sun, J., Mao, Y., Cui, L., Cao, Y., Li, Z., Ling, M., et al. (2021). Using a safe and effective fixative to improve the immunofluorescence staining of bacteria. Methods and applications in fluorescence, 9(3), 035001. https://doi.org/10.1088/2050-6120/abf81e.
[52] Sun, J., Ning, X., Cui, L., Qin, W., Wang, W., and He, S. (2024). Sensitive detection of aflatoxin B1 in foods by aptasensing-based qPCR. Food chemistry, 432, 137240. https://doi.org/10.1016/j.foodchem.2023.137240.
[53] Wang, J., Cao, Y., Li, Z., Dong, M., Dou, W., Xu, X., et al. (2023). Bridge-DNA synthesis triggered by an allosteric aptamer for the colorimetric detection of pathogenic bacteria. Analytical methods : advancing methods and applications, 15(3), 275-283. https://doi.org/10.1039/d2ay01844f.
[54] Zheng, Y., Sun, J., Li, X., Qiu, H., Liao, L., and He, S. (2025). Cell viability assessment by using GelRed/SYTO 9-based double staining. Methods and applications in fluorescence, 13(2), 025001. https://doi.org/10.1088/2050-6120/adaa4e.
[55] Li, Z., Fu, J., Wang, L., Zhou, Y., Li, J., and He, S. (2024). Transformation of proteins into reproductive DNA templates for sensitive quantification of PSA. Talanta, 267, 125206. https://doi.org/10.1016/j.talanta.2023.125206.
[56] Ling, M., Luo, N., Cui, L., Cao, Y., Ning, X., Sun, J., et al. (2022). On-bead DNA synthesis triggered by allosteric probe for detection of carcinoembryonic antigen. Mikrochimica acta, 189(8), 305. https://doi.org/10.1007/s00604-022-05404-4.
[57] Hsu, M.-T., Wang, Y.-K., and Tseng, Y.J. (2022). Exosomal proteins and lipids as potential biomarkers for lung cancer diagnosis, prognosis, and treatment. Cancer, 14(3), 732. https://doi.org/10.3390/cancers14030732.
[58] Shen, H., Cao, D., and Zhang, X. (2023). Advances in exosome research in the management of lung cancer. Current topics in medicinal chemistry, 23(10), 921-930. https://doi.org/10.2174/1568026623666230504101208.
[59] Lee, C.H., Kim, M.H., Lee, M.K., Kim, A., and Son, H.I. (2017). Diagnostic value of microRNAs derived exosomes from bronchoalveolar lavage fluid in early stage lung adenocarcinoma: A pilot study. Virchows Archiv: An International Journal of Pathology, 471, S109-S109.
[60] Tamiya, H., Mitani, A., Saito, A., Ishimori, T., Saito, M., Isago, H., et al. (2018). Exosomal microRNA expression profiling in patients with lung adenocarcinoma-associated malignant pleural effusion. Anticancer research, 38(12), 6707-6714. https://doi.org/10.21873/anticanres.13039.
[61] Thuya, W.L., Peyper, J.M., Myen, T.T., Anuar, N.D., Anwar, A., Gudimella, R., et al. (2024). Exosome autoantibody biomarkers for detection of lung cancer. Military Medical Research, 11(1), 71. https://doi.org/10.1186/s40779-024-00575-y.
[62] Zhang, Y., Liu, W., Zhang, H., Sun, B., Chen, T., Hu, M., et al. (2023). Extracellular vesicle long RNA markers of early-stage lung adenocarcinoma. International journal of cancer, 152(7), 1490-1500. https://doi.org/10.1002/ijc.34386.
[63] Zheng, D., Yang, Y., Wu, C., Wang, H., Zhang, J., Liu, S., et al. (2020). A panel of plasma exosomal miRNAs as diagnosis biomarker to distinguish benign and malignant nodules in non-small cell lung cancer. Cancer Research, 80(16), 756. https://doi.org/10.1158/1538-7445.AM2020-756.
[64] Patel, A.N., and Srinivasan, K. (2025). Deep learning paradigms in lung cancer diagnosis: A methodological review, open challenges, and future directions. Physica medica, 131, 104914. https://doi.org/10.1016/j.ejmp.2025.104914.
[65] Thanoon, M.A., Zulkifley, M.A., Zainuri, M.A.A.M., and Abdani, S.R. (2023). A review of deep learning techniques for lung cancer screening and diagnosis based on CT images. Diagnostics, 13(16), 2617. https://doi.org/10.3390/diagnostics13162617.
[66] Wang, L. (2022). Deep learning techniques to diagnose lung cancer. Cancers, 14(22), 5569. https://doi.org/10.3390/cancers14225569.
[67] Ardila, D., Kiraly, A.P., Bharadwaj, S., Choi, B., Reicher, J.J., Peng, L., et al. (2019). End-to-end lung cancer screening with three-dimensional deep learning on low-dose chest computed tomography. Nature medicine, 25(6), 954-961. https://doi.org/10.1038/s41591-019-0447-x.
[68] Yu, Y., Zeng, D., Ou, Q., Liu, S., Li, A., Chen, Y., et al. (2019). Association of survival and immune-related biomarkers with immunotherapy in patients with non-small cell lung cancer a meta-analysis and individual patient-level analysis. JAMA Network Open, 2(7), e196879. https://doi.org/10.1001/jamanetworkopen.2019.6879.
[69] Lu, H., Wang, H., Zhang, Q., Yoon, S.W., and Won, D. (2019). A 3D convolutional neural network for volumetric image semantic segmentation. Procedia Manufacturing, 39, 422-428.
[70] Alakwaa, W., Nassef, M., and A., B. (2017). Lung cancer detection and classification with 3D convolutional neural network (3D-CNN). International Journal of Advanced Computer Science and Applications (ijacsa), 8(8), 409-417. https://doi.org/10.14569/IJACSA.2017.080853.
[71] Alzubaidi, L., Al-Amidie, M., Al-Asadi, A., Humaidi, A.J., Al-Shamma, O., Fadhel, M.A., et al. (2021). Novel transfer learning approach for medical imaging with limited labeled data. Cancers, 13(7), 1590. https://doi.org/10.3390/cancers13071590.
[72] Sajja, T.K., Devarapalli, R.M., and Kalluri, H.K. (2019). Lung cancer detection based on CT scan images by using deep transfer learning. Traitement du Signal, 36(4), 339-344. https://doi.org/10.18280/ts.360406.
[73] Alom, M.Z., Hasan, M., Yakopcic, C., Taha, T.M., and Asari, V.K. (2021). Inception recurrent convolutional neural network for object recognition. Machine Vision and Applications, 32(1), 28. https://doi.org/10.1007/s00138-020-01157-3.
[74] Kanjee, Z., Crowe, B., and Rodman, A. (2023). Accuracy of a generative artiffcial intelligence model in a complex diagnostic challenge. JAMA, 330(1), 78-80.
[75] Yang, J., Cai, X., Dai, X., and Xie, C. (2025). Assessing the performance of ChatGPT-4 and ChatGPT-4o in lung cancer diagnoses. Journal of translational medicine, 23(1), 346. https://doi.org/10.1186/s12967-025-06337-1.
[76] Kim, S.H., Kim, M.-H., Lee, M.K., and Eom, J.S. (2023). Problems in the pathologic diagnosis of suspected lung cancer. Tuberculosis and respiratory diseases, 86(3), 176-182. https://doi.org/10.4046/trd.2022.0142.
[77] Murakami, S., Yokose, T., Nemoto, D., Suzuki, M., Usui, R., Nakahara, Y., et al. (2021). Suitability of bronchoscopic biopsy tissue samples for next-generation sequencing. Diagnostics, 11(3), 391. https://doi.org/10.3390/diagnostics11030391.
[78] Kage, H., Kohsaka, S., Shinozaki-Ushiku, A., Hiraishi, Y., Sato, J., Nagayama, K., et al. (2019). Small lung tumor biopsy samples are feasible for high quality targeted next generation sequencing. Cancer science, 110(8), 2652-2657. https://doi.org/10.1111/cas.14112.
[79] Feng, J., Zhang, P., Wang, D., Li, Y., and Tan, J. (2024). New strategies for lung cancer diagnosis and treatment: applications and advances in nanotechnology. Biomarker research, 12(1), 136. https://doi.org/10.1186/s40364-024-00686-7.
[80] Kaur, M., Guleria, P., and Kumar, V. (2023). Nanomaterials for diagnosis and treatment of lung cancer: a review of recent patents. Recent patents on anti-cancer drug discovery, 18(2), 114-124. https://doi.org/10.2174/1574892817666220629104641.
Published
Data Availability Statement
None.
Issue
Section
License
Copyright (c) 2026 Cell Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.



