Gene Therapy for Non-Syndromic Hearing Loss
DOI:
https://doi.org/10.71321/703b4949Keywords:
non-syndromic hearing loss, gene therapy, OTOF, DFNB9Abstract
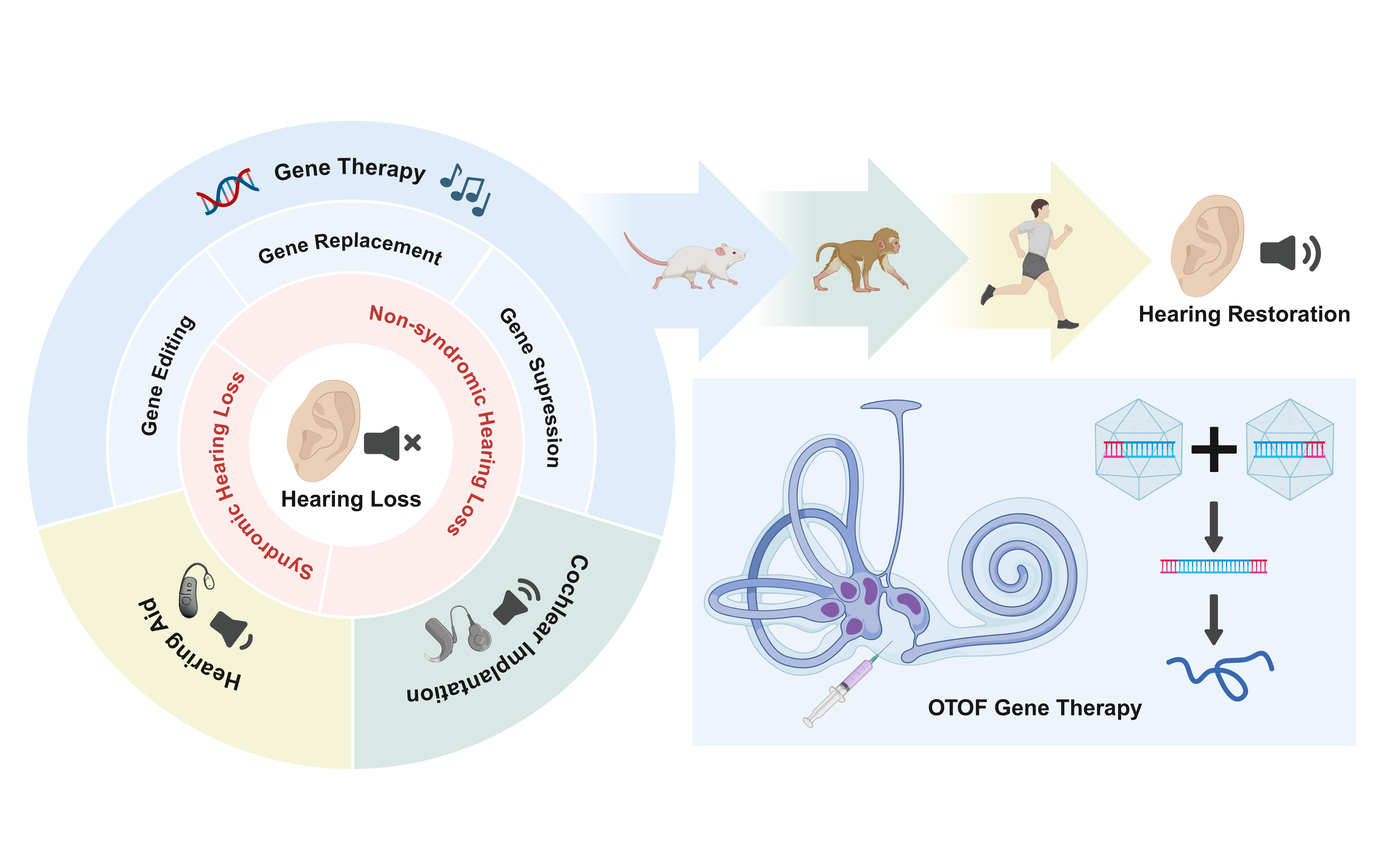
Hereditary hearing loss accounts for over 60% of congenital deafness cases, with non-syndromic hearing loss (NSHL) representing the most common subtype. Typically caused by monogenic mutations, NSHL presents a promising candidate for gene therapy. Recent advances in deciphering the genetic underpinnings of deafness and developing gene delivery systems have greatly accelerated the progress of inner ear gene therapy, leading to a number of breakthrough achievements. This review provides an overview of the latest developments in gene therapy for NSHL. After outlining the genetic basis of NSHL, we summarize the preclinical progress made during the first decade of hereditary deafness gene therapy. Special emphasis is placed on gene replacement strategies for DFNB9, an autosomal recessive form of hearing loss caused by mutations in the OTOF gene. We highlight the remarkable journey of OTOF gene therapy and discuss future directions in this transformative field.
References
[1] Haile LM, Kamenov K, Briant PS, Orji AU, Steinmetz JD, Abdoli A, et al. (2021). Hearing loss prevalence and years lived with disability, 1990–2019: findings from the Global Burden of Disease Study 2019. Lancet, 397(10278), 996-1009. https://doi.org/10.1016/S0140-6736(21)00516-X
[2] Prasad K, Borre ED, Dillard LK, Ayer A, Der C, Bainbridge KE, et al. (2024). Priorities for hearing loss prevention and estimates of global cause-specific burdens of hearing loss: a systematic rapid review. Lancet Glob Health, 12(2), e217-e225. https://doi.org/10.1016/S2214-109X(23)00514-4
[3] Lieu JEC, Kenna M, Anne S, & Davidson L. (2020). Hearing Loss in Children: A Review. JAMA, 324(21), 2195-2205. https://doi.org/10.1001/jama.2020.17647
[4] Morton Cynthia C, & Nance Walter E. (2006). Newborn Hearing Screening — A Silent Revolution. N Engl J Med, 354(20), 2151-2164. https://doi.org/10.1056/NEJMra050700
[5] Walls WD, Azaiez H, & Smith RJH. Hereditary Hearing Loss Homepage. Retrieved 2025.11.3 from https://hereditaryhearingloss.org
[6] Rauschecker JP, & Shannon RV. (2002). Sending Sound to the Brain. Science, 295(5557), 1025-1029. https://doi.org/10.1126/science.1067796
[7] Kerber S, & Seeber BU. (2012). Sound localization in noise by normal-hearing listeners and cochlear implant users. Ear Hear, 33(4), 445-457. https://doi.org/10.1097/AUD.0b013e318257607b
[8] Macherey O, & Carlyon RP. (2014). Cochlear implants. Curr Biol, 24(18), R878-R884. https://doi.org/10.1016/j.cub.2014.06.053
[9] Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, & Sadelain M. (2018). Gene therapy comes of age. Science, 359(6372), eaan4672. https://doi.org/10.1126/science.aan4672
[10] Kulkarni JA, Witzigmann D, Thomson SB, Chen S, Leavitt BR, Cullis PR, et al. (2021). The current landscape of nucleic acid therapeutics. Nat Nanotechnol, 16(6), 630-643. https://doi.org/10.1038/s41565-021-00898-0
[11] Ginn SL, Mandwie M, Alexander IE, Edelstein M, & Abedi MR. (2024). Gene therapy clinical trials worldwide to 2023—an update. J Gene Med, 26(8), e3721. https://doi.org/10.1002/jgm.3721
[12] Lv J, Wang H, Cheng X, Chen Y, Wang D, Zhang L, et al. (2024). AAV1-hOTOF gene therapy for autosomal recessive deafness 9: a single-arm trial. Lancet, 403(10441), 2317-2325. https://doi.org/10.1016/S0140-6736(23)02874-X
[13] Qi J, Tan F, Zhang L, Lu L, Zhang S, Zhai Y, et al. (2024). AAV-Mediated Gene Therapy Restores Hearing in Patients with DFNB9 Deafness. Adv Sci (Weinh), 11(11), 2306788. https://doi.org/10.1002/advs.202306788
[14] Wang H, Chen Y, Lv J, Cheng X, Cao Q, Wang D, et al. (2024). Bilateral gene therapy in children with autosomal recessive deafness 9: single-arm trial results. Nat Med, 30(7), 1898-1904. https://doi.org/10.1038/s41591-024-03023-5
[15] Qi J, Zhang L, Lu L, Tan F, Cheng C, Lu Y, et al. (2025). AAV gene therapy for autosomal recessive deafness 9: a single-arm trial. Nat Med, 31(9), 2917-2926. https://doi.org/10.1038/s41591-025-03773-w
[16] Valayannopoulos V, Bance M, Carvalho DS, Greinwald JH, Jr., Harvey SA, Ishiyama A, et al. (2025). DB-OTO Gene Therapy for Inherited Deafness. N Engl J Med, 10.1056/NEJMoa2400521. https://doi.org/10.1056/NEJMoa2400521
[17] Cheng X, Zhong J, Zhang J, Cui C, Jiang L, Liu Y-w, et al. (2025). Gene Therapy vs Cochlear Implantation in Restoring Hearing Function and Speech Perception for Individuals With Congenital Deafness. JAMA Neurol, 82(9), 941-951. https://doi.org/10.1001/jamaneurol.2025.2053
[18] Smith RJH, Bale JF, Jr., & White KR. (2005). Sensorineural hearing loss in children. Lancet, 365(9462), 879-890. https://doi.org/10.1016/S0140-6736(05)71047-3
[19] Writing Group For Practice Guidelines For Diagnosis Treatment Of Genetic Diseases Medical Genetics Branch Of Chinese Medical Association, Yuan H, Dai P, Liu Y, & Yang T. (2020). Clinical practice guidelines for hereditary non-syndromic deafness. Zhonghua Yi Xue Yi Chuan Xue Za Zhi, 37(3), 269-276. https://doi.org/10.3760/cma.j.issn.1003-9406.2020.03.008
[20] McKusick-Nathans Institute of Genetic Medicine, & Johns Hopkins University (Baltimore MD). Online Mendelian Inheritance in Man, OMIM. Retrieved 2025.11.3 from https://omim.org/
[21] Sloan-Heggen CM, Bierer AO, Shearer AE, Kolbe DL, Nishimura CJ, Frees KL, et al. (2016). Comprehensive genetic testing in the clinical evaluation of 1119 patients with hearing loss. Hum Genet, 135(4), 441-450. https://doi.org/10.1007/s00439-016-1648-8
[22] Mu Y, Han M, Li Y, Di H, Li Z, Li H, et al. (2025). Analysis of deafness gene screening results in 15771 newborn cases in Anyang city of Henan. Front Pediatr, Volume 13 - 2025. https://doi.org/10.3389/fped.2025.1645070
[23] Yan D, Xiang G, Chai X, Qing J, Shang H, Zou B, et al. (2017). Screening of deafness-causing DNA variants that are common in patients of European ancestry using a microarray-based approach. PLOS ONE, 12(3), e0169219. https://doi.org/10.1371/journal.pone.0169219
[24] Yan D, Tekin D, Bademci G, Foster J, Cengiz FB, Kannan-Sundhari A, et al. (2016). Spectrum of DNA variants for non-syndromic deafness in a large cohort from multiple continents. Hum Genet, 135(8), 953-961. https://doi.org/10.1007/s00439-016-1697-z
[25] Sheffield AM, & Smith RJH. (2019). The Epidemiology of Deafness. Cold Spring Harb Perspect Med, 9(9). https://doi.org/10.1101/cshperspect.a033258
[26] Petit C, Bonnet C, & Safieddine S. (2023). Deafness: from genetic architecture to gene therapy. Nat Rev Genet, 24(10), 665-686. https://doi.org/10.1038/s41576-023-00597-7
[27] Ma S, Chen X, Wang Y, & Guo Y. (2025). Mechanisms of congenital hearing loss caused by GJB2 gene mutations and current progress in gene therapy. Gene, 946, 149326. https://doi.org/10.1016/j.gene.2025.149326
[28] Yang T, Vidarsson H, Rodrigo-Blomqvist S, Rosengren SS, Enerbäck S, & Smith RJH. (2007). Transcriptional Control of SLC26A4 Is Involved in Pendred Syndrome and Nonsyndromic Enlargement of Vestibular Aqueduct (DFNB4). Am J Hum Genet, 80(6), 1055-1063. https://doi.org/10.1086/518314
[29] Roux I, Safieddine S, Nouvian R, Grati M, Simmler MC, Bahloul A, et al. (2006). Otoferlin, defective in a human deafness form, is essential for exocytosis at the auditory ribbon synapse. Cell, 127(2), 277-289. https://doi.org/10.1016/j.cell.2006.08.040
[30] Aldè M, Cantarella G, Zanetti D, Pignataro L, La Mantia I, Maiolino L, et al. (2023). Autosomal Dominant Non-Syndromic Hearing Loss (DFNA): A Comprehensive Narrative Review. Biomedicines, 11(6). https://doi.org/10.3390/biomedicines11061616
[31] Yasukawa R, Moteki H, Nishio SY, Ishikawa K, Abe S, Honkura Y, et al. (2019). The Prevalence and Clinical Characteristics of TECTA-Associated Autosomal Dominant Hearing Loss. Genes (Basel), 10(10). https://doi.org/10.3390/genes10100744
[32] Lee S-Y, Kim MY, Han JH, Park SS, Yun Y, Jee S-C, et al. (2023). Ramifications of POU4F3 variants associated with autosomal dominant hearing loss in various molecular aspects. Sci Rep, 13(1), 12584. https://doi.org/10.1038/s41598-023-38272-w
[33] Weegerink NJ, Huygen PL, Schraders M, Kremer H, Pennings RJ, & Kunst HP. (2011). Variable degrees of hearing impairment in a Dutch DFNX4 (DFN6) family. Hear Res, 282(1-2), 167-177. https://doi.org/10.1016/j.heares.2011.08.010
[34] Feng H, Huang S, Ma Y, Yang J, Chen Y, Wang G, et al. (2024). Genomic and phenotypic landscapes of X-linked hereditary hearing loss in the Chinese population. Orphanet J Rare Dis, 19(1), 342. https://doi.org/10.1186/s13023-024-03338-z
[35] Andersen EF, Azzariti DR, Babb L, Berg JS, Biesecker LG, Bly Z, et al. (2025). The Clinical Genome Resource (ClinGen): Advancing genomic knowledge through global curation. Genet Med, 27(1). https://doi.org/10.1016/j.gim.2024.101228
[36] Chen C, Shan W, & Guan M-X. (2024). Defective biogenesis of human mitochondrial ribosomes causes sensorineural deafness. Mitochondr Commun, 2, 114-122. https://doi.org/10.1016/j.mitoco.2024.11.001
[37] Fu X, Wan P, Li P, Wang J, Guo S, Zhang Y, et al. (2021). Mechanism and Prevention of Ototoxicity Induced by Aminoglycosides. Front Cell Neurosci, Volume 15 - 2021. https://doi.org/10.3389/fncel.2021.692762
[38] McDermott JH, Wolf J, Hoshitsuki K, Huddart R, Caudle KE, Whirl-Carrillo M, et al. (2022). Clinical Pharmacogenetics Implementation Consortium Guideline for the Use of Aminoglycosides Based on MT-RNR1 Genotype. Clin Pharmacol Ther, 111(2), 366-372. https://doi.org/10.1002/cpt.2309
[39] Giersch ABS, & Morton CC. (2025). Newborn Screening for Deafness/Hard of Hearing in the Genomic Era. Clin Chem, 71(1), 54-60. https://doi.org/10.1093/clinchem/hvae193
[40] Badonyi M, & Marsh JA. (2025). Prevalence of loss-of-function, gain-of-function and dominant-negative mechanisms across genetic disease phenotypes. Nat Commun, 16(1), 8392. https://doi.org/10.1038/s41467-025-63234-3
[41] Jiang L, Wang D, He Y, & Shu Y. (2023). Advances in gene therapy hold promise for treating hereditary hearing loss. Mol Ther, 31(4), 934-950. https://doi.org/10.1016/j.ymthe.2023.02.001
[42] Izumikawa M, Minoda R, Kawamoto K, Abrashkin KA, Swiderski DL, Dolan DF, et al. (2005). Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat Med, 11(3), 271-276. https://doi.org/10.1038/nm1193
[43] Ishimoto S, Kawamoto K, Kanzaki S, & Raphael Y. (2002). Gene transfer into supporting cells of the organ of Corti. Hear Res, 173(1-2), 187-197. https://doi.org/10.1016/s0378-5955(02)00579-8
[44] Akil O, Seal RP, Burke K, Wang C, Alemi A, During M, et al. (2012). Restoration of hearing in the VGLUT3 knockout mouse using virally mediated gene therapy. Neuron, 75(2), 283-293. https://doi.org/10.1016/j.neuron.2012.05.019
[45] Bankoti K, Generotti C, Hwa T, Wang L, O’Malley BW, Jr., & Li D. (2021). Advances and challenges in adeno-associated viral inner-ear gene therapy for sensorineural hearing loss. Mol Ther Methods Clin Dev, 21, 209-236. https://doi.org/10.1016/j.omtm.2021.03.005
[46] Lahlou G, Calvet C, Giorgi M, Lecomte MJ, & Safieddine S. (2023). Towards the Clinical Application of Gene Therapy for Genetic Inner Ear Diseases. J Clin Med, 12(3). https://doi.org/10.3390/jcm12031046
[47] Gadenstaetter AJ, Krumpoeck PE, & Landegger LD. (2025). Inner Ear Gene Therapy: An Overview from Bench to Bedside. Mol Diagn Ther, 29(2), 161-181. https://doi.org/10.1007/s40291-024-00759-1
[48] Wu F, Sambamurti K, & Sha S. (2022). Current Advances in Adeno-Associated Virus-Mediated Gene Therapy to Prevent Acquired Hearing Loss. J Assoc Res Otolaryngol, 23(5), 569-578. https://doi.org/10.1007/s10162-022-00866-y
[49] Klimara MJ, & Smith RJH. (2023). Advances in cochlear gene therapies. Curr Opin Pediatr, 35(6), 631-640. https://doi.org/10.1097/mop.0000000000001273
[50] Yoshimura H, Shibata SB, Ranum PT, & Smith RJH. (2018). Enhanced viral-mediated cochlear gene delivery in adult mice by combining canal fenestration with round window membrane inoculation. Sci Rep, 8(1), 2980. https://doi.org/10.1038/s41598-018-21233-z
[51] Nist-Lund CA, Pan B, Patterson A, Asai Y, Chen T, Zhou W, et al. (2019). Improved TMC1 gene therapy restores hearing and balance in mice with genetic inner ear disorders. Nat Commun, 10(1), 236. https://doi.org/10.1038/s41467-018-08264-w
[52] Wu X, Zhang L, Li Y, Zhang W, Wang J, Cai C, et al. (2021). Gene therapy via canalostomy approach preserves auditory and vestibular functions in a mouse model of Jervell and Lange-Nielsen syndrome type 2. Nat Commun, 12(1), 697. https://doi.org/10.1038/s41467-020-20808-7
[53] Zhao X, Liu H, Liu H, Cai R, & Wu H. (2022). Gene Therapy Restores Auditory Functions in an Adult Vglut3 Knockout Mouse Model. Hum Gene Ther, 33(13-14), 729-739. https://doi.org/10.1089/hum.2022.062
[54] Chang M, & Kanold PO. (2021). Development of Auditory Cortex Circuits. J Assoc Res Otolaryngol, 22(3), 237-259. https://doi.org/10.1007/s10162-021-00794-3
[55] Dai C, Lehar M, Sun DQ, Rvt LS, Carey JP, MacLachlan T, et al. (2017). Rhesus Cochlear and Vestibular Functions Are Preserved After Inner Ear Injection of Saline Volume Sufficient for Gene Therapy Delivery. J Assoc Res Otolaryngol, 18(4), 601-617. https://doi.org/10.1007/s10162-017-0628-6
[56] Andres-Mateos E, Landegger LD, Unzu C, Phillips J, Lin BM, Dewyer NA, et al. (2022). Choice of vector and surgical approach enables efficient cochlear gene transfer in nonhuman primate. Nat Commun, 13(1), 1359. https://doi.org/10.1038/s41467-022-28969-3
[57] Akil O, Dyka F, Calvet C, Emptoz A, Lahlou G, Nouaille S, et al. (2019). Dual AAV-mediated gene therapy restores hearing in a DFNB9 mouse model. Proc Natl Acad Sci U S A, 116(10), 4496-4501. https://doi.org/doi:10.1073/pnas.1817537116
[58] Al‐Moyed H, Cepeda AP, Jung S, Moser T, Kügler S, & Reisinger E. (2019). A dual‐AAV approach restores fast exocytosis and partially rescues auditory function in deaf otoferlin knock‐out mice. EMBO Mol Med, 11(1), e9396. https://doi.org/10.15252/emmm.201809396
[59] Tang H, Wang H, Wang S, Hu SW, Lv J, Xun M, et al. (2023). Hearing of Otof-deficient mice restored by trans-splicing of N- and C-terminal otoferlin. Hum Genet, 142(2), 289-304. https://doi.org/10.1007/s00439-022-02504-2
[60] Qi J, Zhang L, Tan F, Zhang Y, Zhou Y, Zhang Z, et al. (2024). Preclinical Efficacy And Safety Evaluation of AAV-OTOF in DFNB9 Mouse Model And Nonhuman Primate. Adv Sci (Weinh), 11(3), e2306201. https://doi.org/10.1002/advs.202306201
[61] Benamer N, Le Ribeuz H, Felgerolle C, Calvet C, Postal O, Plion B, et al. (2025). Cochlear gene therapy restores hearing and auditory processing in an atypical DFNB9 mouse model. Commun Med (Lond), 5(1), 229. https://doi.org/10.1038/s43856-025-00926-3
[62] Sellon JB, So KS, D'Arcangelo A, Cancelarich S, Drummond MC, Slade PG, et al. (2024). Recovery kinetics of dual AAV-mediated human otoferlin expression. Front Mol Neurosci, 17, 1376128. https://doi.org/10.3389/fnmol.2024.1376128
[63] Sanes Dan H, & Woolley Sarah MN. (2011). A Behavioral Framework to Guide Research on Central Auditory Development and Plasticity. Neuron, 72(6), 912-929. https://doi.org/10.1016/j.neuron.2011.12.005
[64] Lim R, & Brichta AM. (2016). Anatomical and physiological development of the human inner ear. Hear Res, 338, 9-21. https://doi.org/10.1016/j.heares.2016.02.004
[65] The safety, tolerability, and preliminary efficacy of RRG-003 AAV in the treatment of DFNB9 congenital deafness. (2022). https://www.chictr.org.cn/showproj.html?proj=247690.
[66] A Trial of AAVAnc80-hOTOF Gene Therapy in Individuals With Sensorineural Hearing Loss Due to Otoferlin Gene Mutations. (2023). https://clinicaltrials.gov/study/NCT05821959.
[67] A Phase 1/2, Open-Label, Multicenter Trial with a Single Ascending Dose Cohort with Unilateral Intracochlear Injection Followed by a Bilateral Injection Expansion Cohort to Evaluate the Safety, Tolerability, and Efficacy of DB-OTO in Children and Infants with Biallelic hOTOF Mutations. (2023). https://clinicaltrials.gov/study/NCT05788536.
[68] An Investigator Initiated Study Evaluating the Safety, Tolerability, and Efficacy of OTOV101N+OTOV101C Injection in Treating Patients With OTOF Mutation-related Deafness. (2023). https://clinicaltrials.gov/study/NCT05901480.
[69] A Phase I/II, Open-ended, Adaptative, Open Label Dose Escalation and Expansion Clinical Trial to Evaluate the Efficacy and Safety of Unilateral Intracochlear Injection of SENS-501 Using an Injection System in Children with Severe to Profound Hearing Loss Due to Otoferlin Gene Mutations. (2024). https://clinicaltrials.gov/study/NCT06370351.
[70] The safety, tolerability, and efficacy of EA0010 in the treatment of OTOF-deficiency hearing loss. (2024). https://www.chictr.org.cn/showproj.html?proj=247690.
[71] A Study on the Safety, Tolerability, and Preliminary Efficacy of EH002 in the Treatment of DFNB9 Congenital Deafness. (2024). https://clinicaltrials.gov/study/NCT06722170.
Type
Published
Issue
Section
License
Copyright (c) 2026 Head and Neck Diseases Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







