Gut Microbiota Affects Cardiovascular Diseases via Inflammatory Cytokines: a Bidirectional Two-sample Mendelian Randomization Study and Mediation Analysis
DOI:
https://doi.org/10.71321/c18kt054Keywords:
Gut microbiota, Inflammatory Cytokines, Cardiovascular diseases, Mendelian randomizationAbstract
Background: Cardiovascular diseases (CVDs) arise from complex interactions between genetic predisposition and environmental exposures, manifesting as persistent inflammatory disorders. Although accumulating evidence has implicated gut microbiota in the pathogenesis of CVDs, whether this association reflects a causal relationship remains to be firmly established. Therefore, the present study sought to examine the potential causal links between gut microbiota composition, circulating inflammatory cytokines, and susceptibility to CVDs.
Methods: Summary statistics for 196 gut microbiota taxa, 91 cytokines, and 8 CVD subtypes were extracted from the largest published genome-wide association studies (GWAS) to conduct bidirectional two-sample Mendelian randomization (MR) analysis. The inverse variance weighted (IVW) method was used as the primary statistical approach. We investigated the causal associations between gut microbiota and 8 CVD subtypes, including coronary artery disease (CAD), coronary atherosclerosis (CAS), heart failure (HF), stroke, atrial fibrillation (AF), angina pectoris (AP), calcific aortic valve stenosis (CAVS), and aortic aneurysm. Multivariable MR (MVMR) analysis was further performed to explore the potential mediating role of cytokines in the causal pathways from gut microbiota to CVDs.
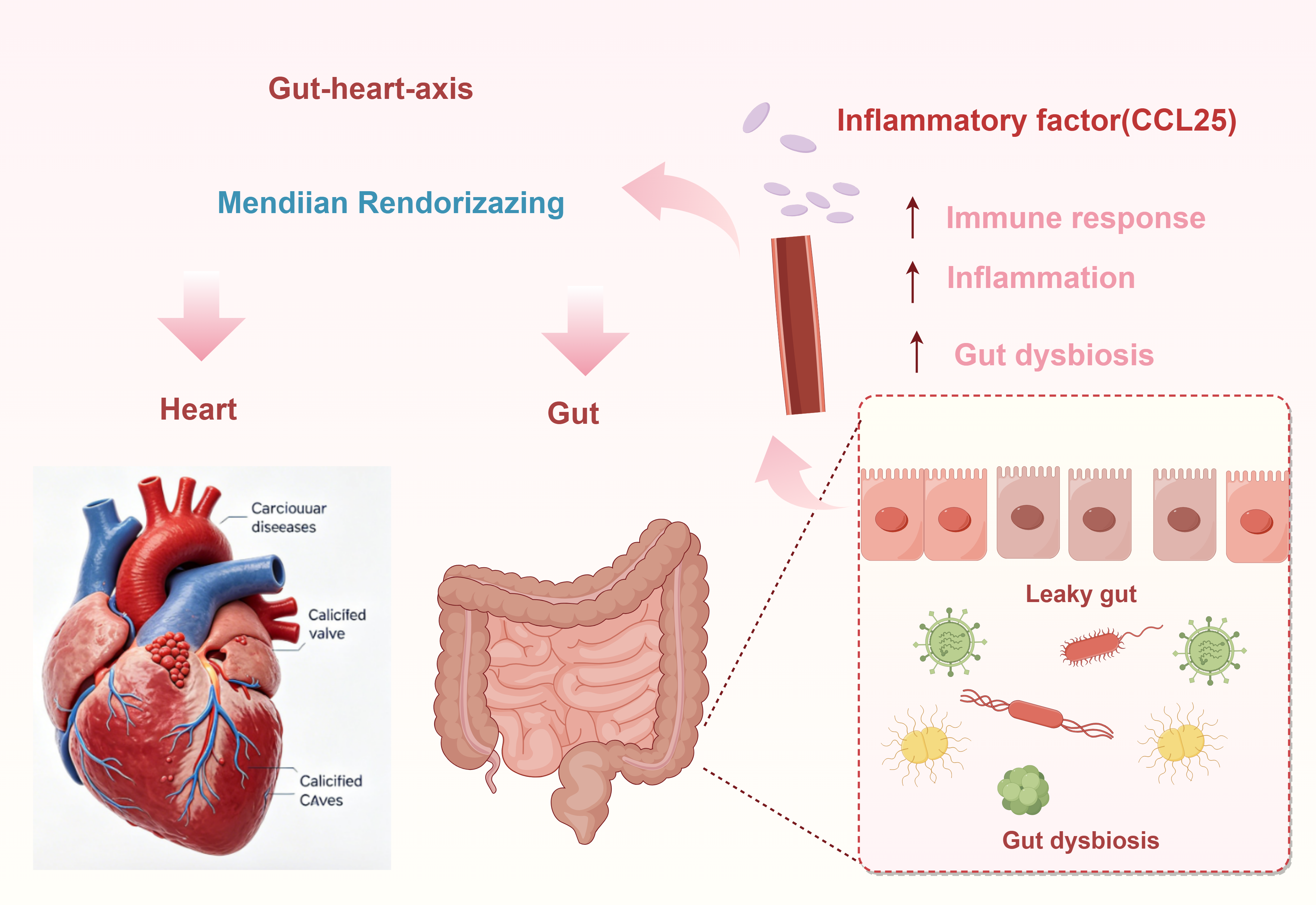
Results: MR analysis identified causal associations between 58 gut microbiota taxa, 46 inflammatory cytokines, and CVDs. Specifically, 34 positive and 24 negative causal effects were observed between gut microbiota and CVDs, along with 16 positive and 30 negative causal effects between cytokines and CVDs. Additionally, mediation analysis identified C-C motif chemokine 25 (CCL25) level as a key mediator in the causal relationship from gut microbiota to CVDs.
Conclusions: Our findings support a causal link between gut microbiota, inflammatory cytokines, and CVDs, with CCL25 mediating the protective effect of specific gut taxa against CAVS. These identified biomarkers offer novel insights into the pathophysiological mechanisms of CVDs and may inform the development of novel strategies for CVD prevention, diagnosis, and treatment.
References
[1] Hemmati M, Kashanipoor S, Mazaheri P, Alibabaei F, Babaeizad A, Asli S, Mohammadi S, Gorgin AH, Ghods K, Yousefi B et al: Importance of gut microbiota metabolites in the development of cardiovascular diseases (CVD). Life sciences 2023, 329:121947.http://dx.doi.org/10.1016/j.lfs.2023.121947
[2] Chen X, Zhang H, Ren S, Ding Y, Remex NS, Bhuiyan MS, Qu J, Tang X: Gut microbiota and microbiota-derived metabolites in cardiovascular diseases. Chinese medical journal 2023, 136(19):2269-2284.http://dx.doi.org/10.1097/cm9.0000000000002206
[3] Jia Q, Li H, Zhou H, Zhang X, Zhang A, Xie Y, Li Y, Lv S, Zhang J: Role and Effective Therapeutic Target of Gut Microbiota in Heart Failure. Cardiovascular therapeutics 2019, 2019:5164298.http://dx.doi.org/10.1155/2019/5164298
[4] Nesci A, Carnuccio C, Ruggieri V, D'Alessandro A, Di Giorgio A, Santoro L, Gasbarrini A, Santoliquido A, Ponziani FR: Gut Microbiota and Cardiovascular Disease: Evidence on the Metabolic and Inflammatory Background of a Complex Relationship. International journal of molecular sciences 2023, 24(10).http://dx.doi.org/10.3390/ijms24109087
[5] Zhang Y, Wang Y, Ke B, Du J: TMAO: how gut microbiota contributes to heart failure. Translational research : the journal of laboratory and clinical medicine 2021, 228:109-125.http://dx.doi.org/10.1016/j.trsl.2020.08.007
[6] Blöbaum L, Witkowski M, Wegner M, Lammel S, Schencke PA, Jakobs K, Puccini M, Reißner D, Steffens D, Landmesser U et al: Intestinal Barrier Dysfunction and Microbial Translocation in Patients with First-Diagnosed Atrial Fibrillation. Biomedicines 2023, 11(1).http://dx.doi.org/10.3390/biomedicines11010176
[7] Lin CH, Chen CC, Chiang HL, Liou JM, Chang CM, Lu TP, Chuang EY, Tai YC, Cheng C, Lin HY et al: Altered gut microbiota and inflammatory cytokine responses in patients with Parkinson's disease. Journal of neuroinflammation 2019, 16(1):129.http://dx.doi.org/10.1186/s12974-019-1528-y
[8] Soysal P, Arik F, Smith L, Jackson SE, Isik AT: Inflammation, Frailty and Cardiovascular Disease. Advances in experimental medicine and biology 2020, 1216:55-64.http://dx.doi.org/10.1007/978-3-030-33330-0_7
[9] Haybar H, Bandar B, Torfi E, Mohebbi A, Saki N: Cytokines and their role in cardiovascular diseases. Cytokine 2023, 169:156261.http://dx.doi.org/10.1016/j.cyto.2023.156261
[10] Gagnon E, Mitchell PL, Manikpurage HD, Abner E, Taba N, Esko T, Ghodsian N, Thériault S, Mathieu P, Arsenault BJ: Impact of the gut microbiota and associated metabolites on cardiometabolic traits, chronic diseases and human longevity: a Mendelian randomization study. Journal of translational medicine 2023, 21(1):60.http://dx.doi.org/10.1186/s12967-022-03799-5
[11] Dai XC, Yu Y, Zhou SY, Yu S, Xiang MX, Ma H: Assessment of the causal relationship between gut microbiota and cardiovascular diseases: a bidirectional Mendelian randomization analysis. BioData mining 2024, 17(1):6.http://dx.doi.org/10.1186/s13040-024-00356-2
[12] Lawlor DA, Harbord RM, Sterne JA, Timpson N, Davey Smith G: Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Statistics in medicine 2008, 27(8):1133-1163.http://dx.doi.org/10.1002/sim.3034
[13] Kurilshikov A, Medina-Gomez C, Bacigalupe R, Radjabzadeh D, Wang J, Demirkan A, Le Roy CI, Raygoza Garay JA, Finnicum CT, Liu X et al: Large-scale association analyses identify host factors influencing human gut microbiome composition. Nature genetics 2021, 53(2):156-165.http://dx.doi.org/10.1038/s41588-020-00763-1
[14] Ahola-Olli AV, Würtz P, Havulinna AS, Aalto K, Pitkänen N, Lehtimäki T, Kähönen M, Lyytikäinen LP, Raitoharju E, Seppälä I et al: Genome-wide Association Study Identifies 27 Loci Influencing Concentrations of Circulating Cytokines and Growth Factors. American journal of human genetics 2017, 100(1):40-50.http://dx.doi.org/10.1016/j.ajhg.2016.11.007
[15] Ji D, Chen WZ, Zhang L, Zhang ZH, Chen LJ: Gut microbiota, circulating cytokines and dementia: a Mendelian randomization study. Journal of neuroinflammation 2024, 21(1):2.http://dx.doi.org/10.1186/s12974-023-02999-0
[16] Jiang S, Yu C, Lv B, He S, Zheng Y, Yang W, Wang B, Li D, Lin J: Two-sample Mendelian randomization to study the causal association between gut microbiota and atherosclerosis. Frontiers in immunology 2023, 14:1282072.http://dx.doi.org/10.3389/fimmu.2023.1282072
[17] Shi Q, Wang Q, Wang Z, Lu J, Wang R: Systemic inflammatory regulators and proliferative diabetic retinopathy: A bidirectional Mendelian randomization study. Frontiers in immunology 2023, 14:1088778.http://dx.doi.org/10.3389/fimmu.2023.1088778
[18] Burgess S, Thompson SG: Avoiding bias from weak instruments in Mendelian randomization studies. International journal of epidemiology 2011, 40(3):755-764.http://dx.doi.org/10.1093/ije/dyr036
[19] Luo S, Li W, Li Q, Zhang M, Wang X, Wu S, Li Y: Causal effects of gut microbiota on the risk of periodontitis: a two-sample Mendelian randomization study. Frontiers in cellular and infection microbiology 2023, 13:1160993.http://dx.doi.org/10.3389/fcimb.2023.1160993
[20] Cai D, Chen J, Wu Y, Jiang C: No causal association between tea consumption and 7 cardiovascular disorders: A two-sample Mendelian randomization study. Frontiers in genetics 2022, 13:989772.http://dx.doi.org/10.3389/fgene.2022.989772
[21] De La Barrera B, Manousaki D: Serum 25-Hydroxyvitamin D Levels and Youth-Onset Type 2 Diabetes: A Two-Sample Mendelian Randomization Study. Nutrients 2023, 15(4).http://dx.doi.org/10.3390/nu15041016
[22] Richmond RC, Hemani G, Tilling K, Davey Smith G, Relton CL: Challenges and novel approaches for investigating molecular mediation. Human molecular genetics 2016, 25(R2):R149-r156.http://dx.doi.org/10.1093/hmg/ddw197
[23] Zheng X, Liu M, Wu Z, Jia Z: Identifying the causal relationship between sedentary behavior and heart failure: Insights from a Mendelian randomization study and mediation analysis. Clinical cardiology 2023, 46(9):1082-1089.http://dx.doi.org/10.1002/clc.24101
[24] Yang J, Nie D, Chen Y, Liu Z, Li M, Gong C, Liu Q: The role of smoking and alcohol in mediating the effect of gastroesophageal reflux disease on lung cancer: A Mendelian randomization study. Frontiers in genetics 2022, 13:1054132.http://dx.doi.org/10.3389/fgene.2022.1054132
[25] Cohen JF, Chalumeau M, Cohen R, Korevaar DA, Khoshnood B, Bossuyt PM: Cochran's Q test was useful to assess heterogeneity in likelihood ratios in studies of diagnostic accuracy. Journal of clinical epidemiology 2015, 68(3):299-306.http://dx.doi.org/10.1016/j.jclinepi.2014.09.005
[26] Burgess S, Thompson SG: Interpreting findings from Mendelian randomization using the MR-Egger method. European journal of epidemiology 2017, 32(5):377-389.http://dx.doi.org/10.1007/s10654-017-0255-x
[27] Zhang Z, Xu W, Zheng Y, Chen C, Kang X, Chen D, Cheng F, Wang X: Causal relationship between psoriasis vulgaris and dementia: Insights from Mendelian randomization analysis. Experimental dermatology 2024, 33(1):e14984.http://dx.doi.org/10.1111/exd.14984
[28] Verbanck M, Chen CY, Neale B, Do R: Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nature genetics 2018, 50(5):693-698.http://dx.doi.org/10.1038/s41588-018-0099-7
[29] Lu W, Cen J, Dai Q, Tao H, Peng L: Gut microbiota does not play a mediating role in the causal association between inflammatory bowel disease and several its associated extraintestinal manifestations: a Mendelian randomization study. Frontiers in immunology 2023, 14:1296889.http://dx.doi.org/10.3389/fimmu.2023.1296889
[30] Zhang Y, Zhang S, Li B, Luo Y, Gong Y, Jin X, Zhang J, Zhou Y, Zhuo X, Wang Z et al: Gut microbiota dysbiosis promotes age-related atrial fibrillation by lipopolysaccharide and glucose-induced activation of NLRP3-inflammasome. Cardiovascular research 2022, 118(3):785-797.http://dx.doi.org/10.1093/cvr/cvab114
[31] Zhao J, Zhang Q, Cheng W, Dai Q, Wei Z, Guo M, Chen F, Qiao S, Hu J, Wang J et al: Heart-gut microbiota communication determines the severity of cardiac injury after myocardial ischaemia/reperfusion. Cardiovascular research 2023, 119(6):1390-1402.http://dx.doi.org/10.1093/cvr/cvad023
[32] Chen W, Zhao Y, Zhao Q, Zhou Y, Ma C, Dong L, Luo Y, Zhang Z, Chen F, Hu X et al: Lachnospiraceae-Derived Extracellular Vesicles Mediate the Cardioprotective Effects of Barley Leaf in Myocardial Infarction by Improving Intestinal Stem Cell Function. Journal of extracellular vesicles 2026, 15(3):e70250.http://dx.doi.org/10.1002/jev2.70250
[33] Zou Y, Song X, Liu N, Sun W, Liu B: Intestinal Flora: A Potential New Regulator of Cardiovascular Disease. Aging and disease 2022, 13(3):753-772.http://dx.doi.org/10.14336/ad.2021.1022
[34] Liu Q, Li Y, Song X, Wang J, He Z, Zhu J, Chen H, Yuan J, Zhang X, Jiang H et al: Both gut microbiota and cytokines act to atherosclerosis in ApoE-/- mice. Microbial pathogenesis 2020, 138:103827.http://dx.doi.org/10.1016/j.micpath.2019.103827
[35] Georgakis MK, Malik R, Burgess S, Dichgans M: Additive Effects of Genetic Interleukin-6 Signaling Downregulation and Low-Density Lipoprotein Cholesterol Lowering on Cardiovascular Disease: A 2×2 Factorial Mendelian Randomization Analysis. Journal of the American Heart Association 2022, 11(1):e023277.http://dx.doi.org/10.1161/jaha.121.023277
[36] Al Bander Z, Nitert MD, Mousa A, Naderpoor N: The Gut Microbiota and Inflammation: An Overview. International journal of environmental research and public health 2020, 17(20).http://dx.doi.org/10.3390/ijerph17207618
[37] Chen Y, Zeng L: Peripheral Inflammatory Factors and Acute Myocardial Infarction Risk: A Mendelian Randomization Study. Global heart 2023, 18(1):55.http://dx.doi.org/10.5334/gh.1269
[38] Ridker PM, Tuttle KR, Perkovic V, Libby P, MacFadyen JG: Inflammation drives residual risk in chronic kidney disease: a CANTOS substudy. European heart journal 2022, 43(46):4832-4844.http://dx.doi.org/10.1093/eurheartj/ehac444
[39] Müller FS, Aherrahrou Z, Grasshoff H, Heidorn MW, Humrich JY, Johanson L, Aherrahrou R, Reinberger T, Schulz A, Ten Cate V et al: Autoantibodies against the chemokine receptor 3 predict cardiovascular risk. European heart journal 2023, 44(47):4935-4949.http://dx.doi.org/10.1093/eurheartj/ehad666
[40] Lv J, Wang J, Yu Y, Zhao M, Yang W, Liu J, Zhao Y, Yang Y, Wang G, Guo L et al: Alterations of gut microbiota are associated with blood pressure: a cross-sectional clinical trial in Northwestern China. Journal of translational medicine 2023, 21(1):429.http://dx.doi.org/10.1186/s12967-023-04176-6
[41] Valassi E, Manichanh C, Amodru V, Fernández PG, Gaztambide S, Yañez F, Martel-Duguech L, Puig-Domingo M, Webb SM: Gut microbial dysbiosis in patients with Cushing's disease in long-term remission. Relationship with cardiometabolic risk. Frontiers in endocrinology 2023, 14:1074757.http://dx.doi.org/10.3389/fendo.2023.1074757
[42] Lambeth SM, Carson T, Lowe J, Ramaraj T, Leff JW, Luo L, Bell CJ, Shah VO: Composition, Diversity and Abundance of Gut Microbiome in Prediabetes and Type 2 Diabetes. Journal of diabetes and obesity 2015, 2(3):1-7.http://dx.doi.org/10.15436/2376-0949.15.031
[43] Karlsson FH, Fåk F, Nookaew I, Tremaroli V, Fagerberg B, Petranovic D, Bäckhed F, Nielsen J: Symptomatic atherosclerosis is associated with an altered gut metagenome. Nature communications 2012, 3:1245.http://dx.doi.org/10.1038/ncomms2266
[44] Liu Y, Liu Y, Deng J, Li W, Nie X: Fibroblast Growth Factor in Diabetic Foot Ulcer: Progress and Therapeutic Prospects. Frontiers in endocrinology 2021, 12:744868.http://dx.doi.org/10.3389/fendo.2021.744868
[45] Sinha SK, Miikeda A, Fouladian Z, Mehrabian M, Edillor C, Shih D, Zhou Z, Paul MK, Charugundla S, Davis RC et al: Local M-CSF (Macrophage Colony-Stimulating Factor) Expression Regulates Macrophage Proliferation and Apoptosis in Atherosclerosis. Arteriosclerosis, thrombosis, and vascular biology 2021, 41(1):220-233.http://dx.doi.org/10.1161/atvbaha.120.315255
[46] Mishra V, Sinha SK, Rajavashisth TB: Role of macrophage colony-stimulating factor in the development of neointimal thickening following arterial injury. Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology 2016, 25(4):284-292.http://dx.doi.org/10.1016/j.carpath.2016.04.003
[47] Rabe T, Lazar K, Cambronero C, Goelz R, Hamprecht K: Human Cytomegalovirus (HCMV) Reactivation in the Mammary Gland Induces a Proinflammatory Cytokine Shift in Breast Milk. Microorganisms 2020, 8(2).http://dx.doi.org/10.3390/microorganisms8020289
[48] Chang TT, Chen JW: Emerging role of chemokine CC motif ligand 4 related mechanisms in diabetes mellitus and cardiovascular disease: friends or foes? Cardiovascular diabetology 2016, 15(1):117.http://dx.doi.org/10.1186/s12933-016-0439-9
[49] Zhang Y, Ye Y, Tang X, Wang H, Tanaka T, Tian R, Yang X, Wang L, Xiao Y, Hu X et al: CCL17 acts as a novel therapeutic target in pathological cardiac hypertrophy and heart failure. The Journal of experimental medicine 2022, 219(8).http://dx.doi.org/10.1084/jem.20200418
Type
Published
Data Availability Statement
All GWAS summary statistics used in this study are publicly available from the IEU Open GWAS database, FinnGen database, and MiBioGen consortium, with detailed sources specified in the manuscript. The full analytical code can be obtained from the corresponding author upon reasonable request.
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







