High-risk Factors and a Nomogram Prediction Model for Pulmonary Fungal Infection in Elderly Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease
DOI:
https://doi.org/10.71321/d3zf3339Keywords:
elderly acute exacerbation of chronic obstructive pulmonary disease, fungal infection, risk factors, nomogram, prediction modelAbstract
Background: Pulmonary fungal infection is a major risk factor for death and prolonged hospitalization in elderly patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD). At present, early recognition of these infections remains difficult. This study therefore collected clinical data to develop an early-prediction model for fungal pneumonia complicating AECOPD.
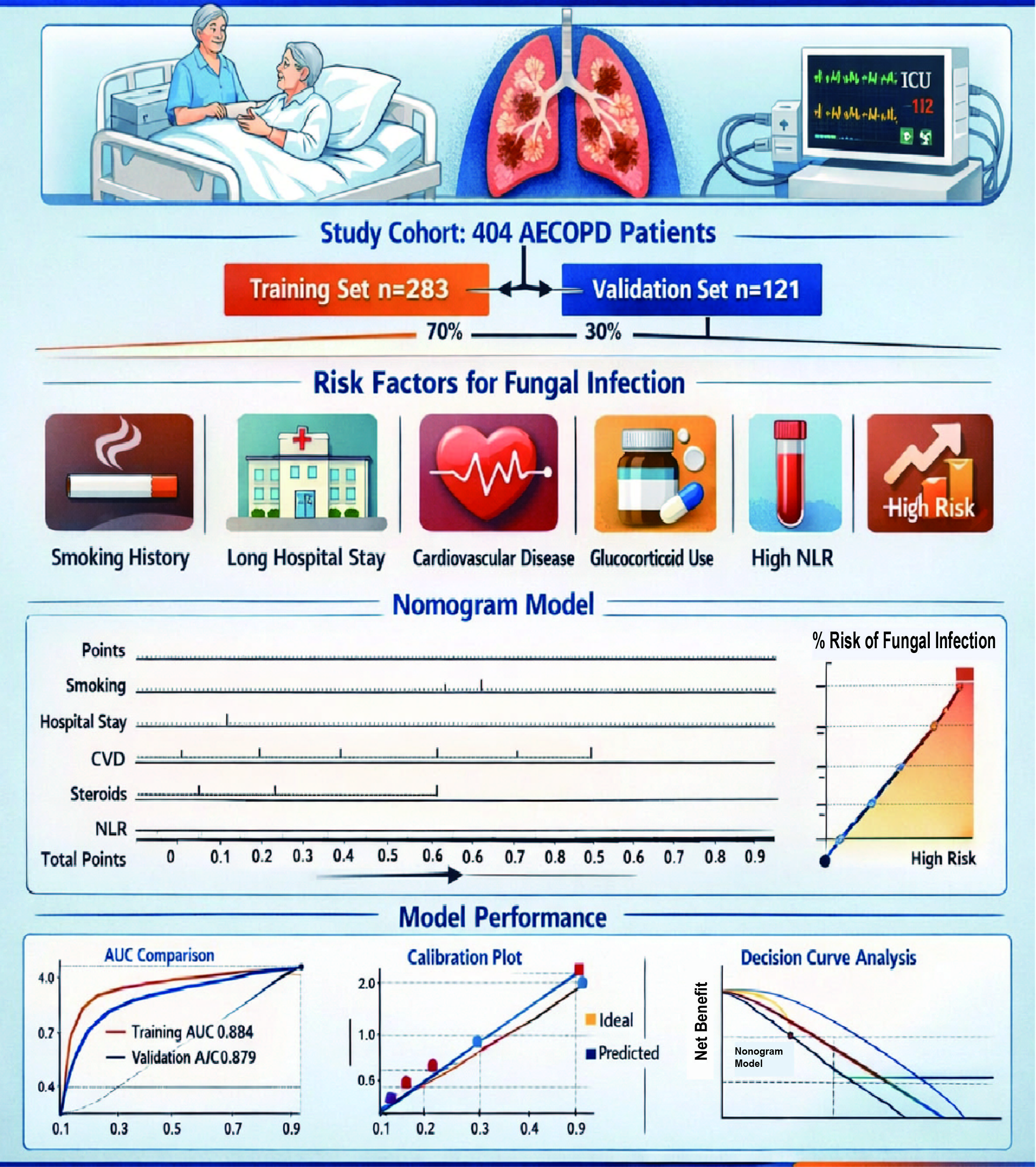
Methods: We enrolled 404 elderly AECOPD patients admitted to the Department of Respiratory Medicine, First Affiliated Hospital of Anhui Medical University, from January to December 2023. The cohort was randomly split 7:3 into a training set (n =283) and a validation set (n =121). Univariate logistic regression was first performed in the training set; variables with p < 0.05 were entered into a multivariate model. Significant factors were used to construct a nomogram. Model performance was evaluated in both sets by the area under the receiver operating characteristic curve (AUC), calibration plots, and decision-curve analysis (DCA).
Results: Smoking history, length of hospital stay, concomitant cardiovascular disease, glucocorticoid use, and the neutrophil-to-lymphocyte ratio (NLR) were independent risk factors for fungal infection during AECOPD (p < 0.05). The nomogram achieved AUCs of 0.884[95% CI: 0.838–0.930] in the training set and 0.879 [95% CI: 0.795–0.962] in the validation set. Calibration and DCA curves indicated good clinical utility.
Conclusion: Smoking history, prolonged hospitalization, concurrent cardiovascular disease, glucocorticoid therapy, and elevated NLR are independent risk factors for fungal infection in AECOPD. The constructed nomogram exhibits strong predictive performance.
References
[1] Maitre T, Cottenet J, Godet C, Roussot A, Abdoul Carime N, Ok V, et al. (2021). Chronic pulmonary aspergillosis: prevalence, favouring pulmonary diseases and prognosis. Eur Respir J, 58(2). https://doi.org/10.1183/13993003.03345-2020
[2] Wang YY, Wang J, Lu ZW, Zhou QQ, Cao YG, Du YJ, et al. (2025). Global, regional, and national burden of lower respiratory infections and chronic obstructive pulmonary disease, 1990-2021: a systematic analysis from the global burden of disease study 2021. Infection, 53(5), 2191-2202. https://doi.org/10.1007/s15010-025-02566-0
[3] Liu SX, M P, W L, QZ H, GF H, MX W, et al. (2023). Global, regional, and national burden of lower respiratory infections and chronic obstructive pulmonary disease. Chinese Journal of Gerontology, 43(04), 817-821. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y6vbIErhuHCGdSON2euxkNxtOJ15W8ZKsIb6m0jGmg8hRXcJJPD3v2zVwFUOIC0-_pwdUd446TsZLSCFe04wlPmp2pfioFmHKaSLEOOitUnRwTvimSutigfjRYHnWQKgY1xAFrrt73OB4feNgGjfAY-50JRDvUA9ThERWVwhjCN1wu2cdwyo0Ze&uniplatform=NZKPT&language=CHS
[4] Leung JM, Tiew PY, Mac Aogáin M, Budden KF, Yong VF, Thomas SS, et al. (2017). The role of acute and chronic respiratory colonization and infections in the pathogenesis of COPD. Respirology, 22(4), 634-650. https://doi.org/10.1111/resp.13032
[5] Shen H, Wu HB, & Y C. (2023). Risk factors for pulmonary fungal infection in elderly patients with acute exacerbation of chronic obstructive pulmonary disease. Chinese Journal of Gerontology, 43(19), 4687-4690. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y7s60nL-dyCsSQ3zFSN3lMmQDsCdZhcjfg3aYk_bUhx5PGIz5riDPrLP9cl_Mj3FgQrS4iAuqpNFwNS4qVkcdVOMIykzo6P339lfOARWrQ2NTKmmUyUoxxH0fDqnAUvuQTcSeVhGt2E-ASM7gT9eeiNzraC4F4zcsZG4dhnDMWZvR1pr8l25Uvi&uniplatform=NZKPT&language=CHS
[6] Yu YL, Lin CK, Zhu CS, & Y W. (2024). Development of a nomogram-based risk prediction model and web calculator for acute exacerbation of chronic obstructive pulmonary disease. Journal of Practical Cardio-Cerebro-Pulmonary Vascular Disease, 32(06), 16-21. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y7GzGDW0xORGsVFn1liQ2LEipg-07rgnn7dtMHF_awAH2cRHg6piewzFEXsYO87oEkxkIftN7uBIEtvKxVypTlhX68nfqjFKhRoOekPtMYRPaCBdyUj3gGRiw7EsIa2duF1iiGJ5C2aLButMfbnqMqDZtOZ8xSDUUSTRyFpdtDdU0MrFSEz08H0&uniplatform=NZKPT&language=CHS
[7] Lin X, & N H. (2024). A nomogram prediction model for secondary pulmonary fungal infection in acute exacerbation of chronic obstructive pulmonary disease. Chinese Journal of Respiratory and Critical Care Medicine, 23(02), 77-85. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y4NvgGQDBl7ny3HJaG6RxQXoN9BYqBQaQII3c4hpOQCXalPCMwxrxjF5wYnQbY-VwTMk_FhG5Ph3tuDzpW_gqxnYMIXKrCUXliYSr9hq2GmyD3nrQtjLKZh3-zRWd3dJ7E2CGDr_CCxpGIUMj_t9aNPWWBY5nLWPkOackiWYzDXAtfro9lpvqto&uniplatform=NZKPT&language=CHS
[8] Expert Panel Consensus on the Diagnosis and Treatment of Pulmonary Fungal Disease. (2007). Chinese Journal of Tuberculosis and Respiratory Diseases, 30(11), 821-834. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y53kDiZwO-TuwoDCoYVkNCo8dD7j5RucNX65emn3Er-YO0fF8t2b-awEBUTv5cxYuJnSKNkQprYgek22lLQAeK0hDzVURmkw1AWnzQ3a7rOKr9HTRuOaSRIZYsEiFpYCP7XEi_G7gg_OTTfu6JhVxs9KIsF1xYruhHKFeU1-Eobbg==&uniplatform=NZKPT&language=CHS
[9] Lugg ST, Scott A, Parekh D, Naidu B, & Thickett DR. (2022). Cigarette smoke exposure and alveolar macrophages: mechanisms for lung disease. Thorax, 77(1), 94-101. https://doi.org/10.1136/thoraxjnl-2020-216296
[10] Lin L, Yi X, Liu H, Meng R, Li S, Liu X, et al. (2023). The airway microbiome mediates the interaction between environmental exposure and respiratory health in humans. Nat Med, 29(7), 1750-1759. https://doi.org/10.1038/s41591-023-02424-2
[11] Razia DEM, Gao C, Wang C, An Y, Wang F, Liu L, et al. (2025). Targeting Non-Eosinophilic Immunological Pathways in COPD and AECOPD: Current Insights and Therapeutic Strategies. Int J Chron Obstruct Pulmon Dis, 20, 511-532. https://doi.org/10.2147/copd.S506616
[12] Fardet L, Petersen I, & Nazareth I. (2016). Common Infections in Patients Prescribed Systemic Glucocorticoids in Primary Care: A Population-Based Cohort Study. PLoS Med, 13(5), e1002024. https://doi.org/10.1371/journal.pmed.1002024
[13] Diaz-Jimenez D, Kolb JP, & Cidlowski JA. (2021). Glucocorticoids as Regulators of Macrophage-Mediated Tissue Homeostasis. Front Immunol, 12, 669891. https://doi.org/10.3389/fimmu.2021.669891
[14] Chen HD, Fu HQ, & YG N. (2024). Development and validation of a nomogram for predicting prognosis in COPD patients complicated with pulmonary infection. Anhui Medical Journal, 45(05), 598-602. https://link.cnki.net/urlid/34.1077.R.20240514.1704.038
[15] Rabe KF, Hurst JR, & Suissa S. (2018). Cardiovascular disease and COPD: dangerous liaisons? Eur Respir Rev, 27(149). https://doi.org/10.1183/16000617.0057-2018
[16] McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, et al. (2014). Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med, 371(11), 993-1004. https://doi.org/10.1056/NEJMoa1409077
[17] Liu Y, Xu J, Ma L, Cao GY, & FD Z. (2024). Association of peripheral blood eosinophils, D-dimer and neutrophil-to-lymphocyte ratio with pulmonary ventilatory function in AECOPD patients. Tianjin Medical Journal, 52(12), 1261-1265. https://link.cnki.net/urlid/12.1116.R.20240929.1023.004
[18] Manolis AA, Manolis TA, Melita H, Mikhailidis DP, & Manolis AS. (2022). Low serum albumin: A neglected predictor in patients with cardiovascular disease. Eur J Intern Med, 102, 24-39. https://doi.org/10.1016/j.ejim.2022.05.004
[19] Y C. (2017). Effect of blood glucose management on pulmonary fungal infection in patients with diabetes mellitus and chronic obstructive pulmonary disease. Diabetes New World, 20(03), 142-144. https://doi.org/10.16658/j.cnki.1672-4062.2017.03.142
[20] Zhang L, Jiang F, Xie Y, Mo Y, Zhang X, & Liu C. (2023). Diabetic endothelial microangiopathy and pulmonary dysfunction. Front Endocrinol (Lausanne), 14, 1073878. https://doi.org/10.3389/fendo.2023.1073878
[21] Han XY, Jin XY, & LH C. (2020). Relationship between high-resolution CT phenotypes and airway inflammation in chronic obstructive pulmonary disease. Journal of Dalian Medical University. Journal of Dalian Medical University, 42(01), 27-31. https://kns.cnki.net/kcms2/article/abstract?v=vQ2wnlBr1y4RWp9v061V9ZG0TTusuymF9nfPhEPQPanIB2o6exgBvxcLm8wQ2jQZXzECXhqsp-J1avxqXS2-CziPNkkOi2Ymu6BGMCAL0G1W8DkIfs6DXKQ0QhZHi9VuiteXW3xXqqSc2zTmilz-EDcVOC69eYSLMJ2izES_mHKVrNHFJVMmHLjA1uLBcyFl&uniplatform=NZKPT&language=CHS
Type
Published
Data Availability Statement
Not applicable.
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







