Risk Factors of Reactive Cutaneous Capillary Endothelial Proliferation in Advanced Non-small Cell Lung Cancer Treated with Camrelizumab
DOI:
https://doi.org/10.71321/ncbcdv45Keywords:
Camrelizumab, advanced non-small cell lung cancer (NSCLC), reactive cutaneous capillary endothelial proliferation (RCCEP), albumin, platelet, anti-angiogenic drugsAbstract
Background: Reactive cutaneous capillary endothelial proliferation (RCCEP) is the most common cutaneous immune-related adverse event (irAE) of camrelizumab. The purpose of our research was to explore the risk factors associated with RCCEP and their predictive values.
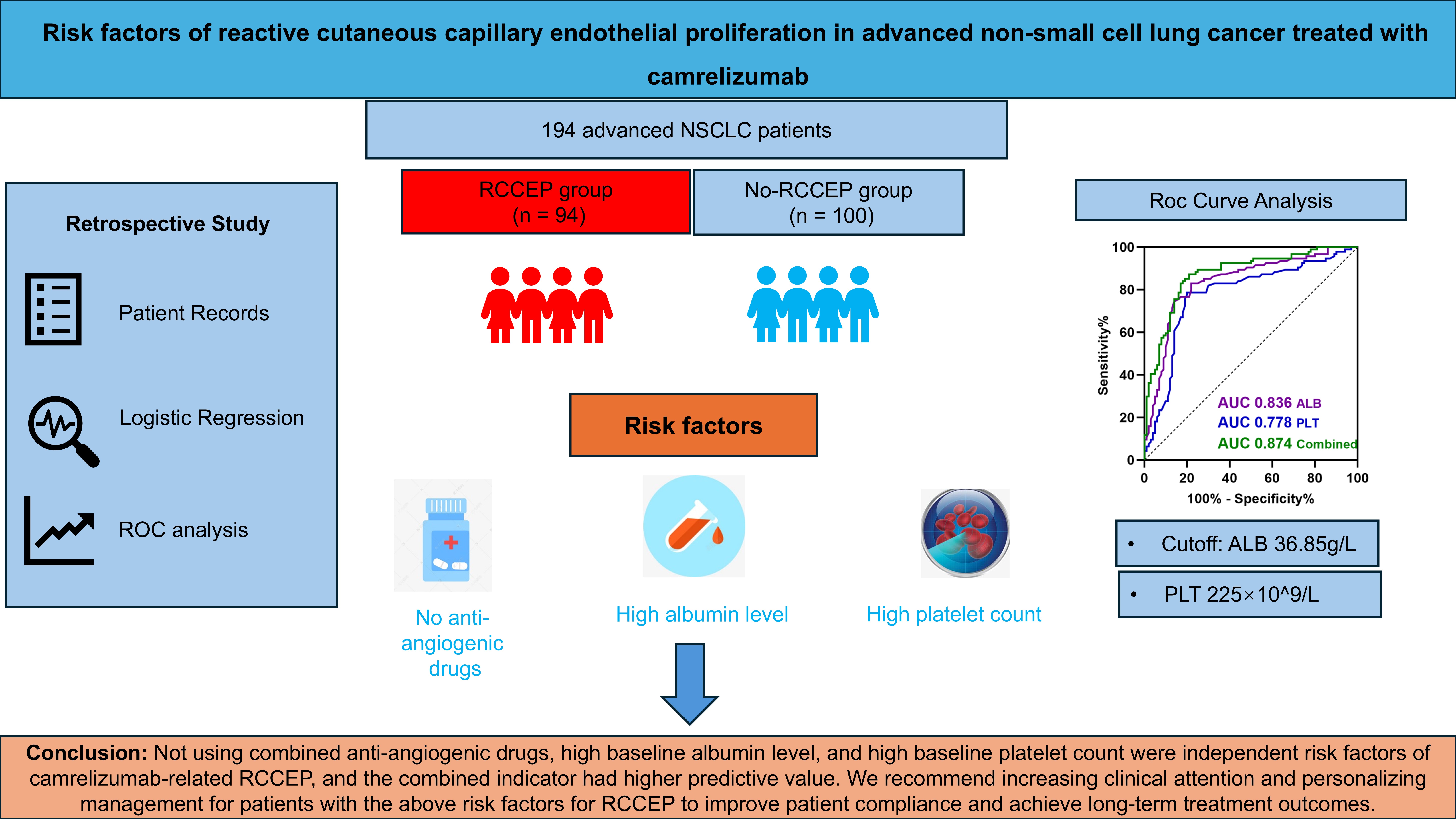
Methods: We conducted a retrospective investigation examining clinical records of advanced non-small cell lung cancer (NSCLC) patients treated with camrelizumab at our hospital between June 2022 and December 2024. Study participants were grouped based on whether they developed RCCEP. To pinpoint potential risk factors associated with RCCEP, we employed logistic regression analysis, while receiver operating characteristic (ROC) curve analysis helped determine the predictive values of our identified variables.
Results: Among 194 patients, 94 (48.5%) developed RCCEP, of whom 92 cases developed grade 1-2 RCCEP, only 2 cases developed grade 3 RCCEP, with no grade 4-5 cases. Multivariate logistic regression analysis suggested that not using combined anti-angiogenic drugs (OR: 2.962, 95% CI 1.042-8.422, P = 0.042), high baseline albumin level (OR: 1.422, 95% CI 1.264-1.599, P < 0.001), and high baseline platelet count (OR: 1.018, 95% CI 1.010-1.026, P < 0.001) were significantly associated with RCCEP. ROC curve analysis showed that baseline albumin level, baseline platelet count, and their combination predicted RCCEP occurrence in advanced NSCLC patients receiving camrelizumab, with areas under the curve (AUCs) of 0.836 (95% CI 0.777-0.895), 0.778 (95% CI 0.709-0.847), and 0.874 (95% CI 0.823-0.924), respectively. The cutoff values for albumin and platelet were calculated using the maximum Youden index from the ROC curve, which were 36.85 g/L and 225×109/L, respectively.
Conclusion: Not using combined anti-angiogenic drugs, high baseline albumin level, and high baseline platelet count were independent risk factors of camrelizumab-related RCCEP, and the combined indicator had higher predictive value. We recommend increasing clinical attention and personalizing management for patients with the above risk factors for RCCEP to improve patient compliance and achieve long-term treatment outcomes.
References
[1] Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clinicians. 2024 May;74(3):229–63. https://doi.org/10.3322/caac.21834
[2] Zhou X, Yao Z, Yang H, Liang N, Zhang X, Zhang F. Are immune-related adverse events associated with the efficacy of immune checkpoint inhibitors in patients with cancer? A systematic review and meta-analysis. BMC Med. 2020 Dec;18(1):87. https://doi.org/10.1186/s12916-020-01549-2
[3] Johnson DB, Nebhan CA, Moslehi JJ, Balko JM. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol. 2022 Apr;19(4):254–67. https://doi.org/10.1038/s41571-022-00600-w
[4] Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020 May 7;6(1):38. https://doi.org/10.1038/s41572-020-0160-6
[5] Naidoo J, Page DB, Li BT, Connell LC, Schindler K, Lacouture ME, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies. Annals of Oncology. 2015 Dec;26(12):2375–91. https://doi.org/10.1093/annonc/mdv383
[6] Chen X, Ma L, Wang X, Mo H, Wu D, Lan B, et al. Reactive capillary hemangiomas: a novel dermatologic toxicity following anti-PD-1 treatment with SHR-1210. Cancer Biol Med. 2019;16(1):173–81. https://doi.org/10.20892/j.issn.2095-3941.2018.0172
[7] Wang F, Qin S, Sun X, Ren Z, Meng Z, Chen Z, et al. Reactive cutaneous capillary endothelial proliferation in advanced hepatocellular carcinoma patients treated with camrelizumab: data derived from a multicenter phase 2 trial. J Hematol Oncol. 2020;13(1). https://doi.org/10.1186/s13045-020-00886-2
[8] Wang H, Xia J, Cao M, Zhao Y, Yu A, Qin X, et al. Observation on the Therapeutic Efficacy of Camrelizumab Combined with Chemotherapy in Non-small Cell Lung Cancer and the Cutaneous Immune-related Adverse Events: A Retrospective Study. ACAMC. 2025 May;25(8):574–87. https://doi.org/10.2174/0118715206350978241105080452
[9] Qu W, Wang F, Qin S, Sun Y, Huang C. Reactive cutaneous capillary endothelial proliferation following camrelizumab monotherapy or combination therapy for multi-cancers: a large-scale pooled analysis of 10 studies in China. Ther Adv Med Oncol. 2024 Jan;16:17588359241242607. https://doi.org/10.1177/17588359241242607
[10] Hwang SJE, Carlos G, Wakade D, Byth K, Kong BY, Chou S, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: A single-institution cohort. Journal of the American Academy of Dermatology. 2016 Mar;74(3):455-461.e1. https://doi.org/10.1016/j.jaad.2015.10.029
[11] Song G, Zhang F, Cheng H. Thalidomide for prevention of camrelizumab‐induced reactive cutaneous capillary endothelial proliferation. Aust J Dermatology. 2022 May;63(2):217–21. https://doi.org/10.1111/ajd.13812
[12] Hu WT, Zhang Q, Zhang Z, He X, Zhou M, Guo Y, et al. Eosinophil and IFN-γ associated with immune-related adverse events as prognostic markers in patients with non-small cell lung cancer treated with immunotherapy. Front Immunol. 2023 Mar 6;14:1112409. https://doi.org/10.3389/fimmu.2023.1112409
[13] Zhang X, Zhang B, Li D, Yang Y, Lin S, Zhao R, et al. Peripheral blood cell counts as predictors of immune-related adverse events in cancer patients receiving immune checkpoint inhibitors: a systematic review and meta-analysis. Front Immunol. 2025 Jan 30;16:1528084. https://doi.org/10.3389/fimmu.2025.1528084
[14] Kondo T, Okamoto I, Sato H, Koyama N, Fushimi C, Okada T, et al. Age‐based efficacy and safety of nivolumab for recurrent or metastatic head and neck squamous cell carcinoma: A multicenter retrospective study. Asia-Pac J Clncl Oncology. 2020 Dec;16(6):340–7. https://doi.org/10.1111/ajco.13374
[15] Duma N, Abdel-Ghani A, Yadav S, Hoversten KP, Reed CT, Sitek AN, et al. Sex Differences in Tolerability to Anti-Programmed Cell Death Protein 1 Therapy in Patients with Metastatic Melanoma and Non-Small Cell Lung Cancer: Are We All Equal? The Oncologist. 2019 Nov 1;24(11):e1148–55. https://doi.org/10.1634/theoncologist.2019-0094
[16] He X, Fang J, Yu P, Hu W, Zhang Q, Zhang Z, et al. Risk of reactive cutaneous capillary endothelial proliferation induced by camrelizumab in patients with non-small cell lung cancer: a retrospective study. J Thorac Dis. 2023 Dec;15(12):6687–96. https://doi.org/10.21037/jtd-23-1144
[17] Luo HY, Hou ZG, Ma C, Chen Y, Zheng Z, Xu RH. 394P Reactive cutaneous capillary endothelial proliferation in esophageal squamous cell carcinoma patients treated with camrelizumab-based therapy: A pooled analysis of two phase III trials. Annals of Oncology. 2025 Jul;36:S154. https://doi.org/10.1016/j.annonc.2025.05.410
[18] Lickliter JD, Gan HK, Voskoboynik M, Arulananda S, Gao B, Nagrial A, et al. A First-in-Human Dose Finding Study of Camrelizumab in Patients with Advanced or Metastatic Cancer in Australia. DDDT. 2020 Mar;Volume 14:1177–89. https://doi.org/10.2147/DDDT.S243787
[19] Xu J, Shen J, Gu S, Zhang Y, Wu L, Wu J, et al. Camrelizumab in Combination with Apatinib in Patients with Advanced Hepatocellular Carcinoma (RESCUE): A Nonrandomized, Open-label, Phase II Trial. Clinical Cancer Research. 2021 Feb 15;27(4):1003–11. https://doi.org/10.1158/1078-0432.CCR-20-2571
[20] Qu W, Wang F, Qin S, Sun Y, Huang C. Reactive cutaneous capillary endothelial proliferation following camrelizumab monotherapy or combination therapy for multi-cancers: a large-scale pooled analysis of 10 studies in China. Ther Adv Med Oncol. 2024 Jan;16:17588359241242607. https://doi.org/10.1177/17588359241242607
[21] Hata H, Matsumura C, Chisaki Y, Nishioka K, Tokuda M, Miyagi K, et al. A Retrospective Cohort Study of Multiple Immune-Related Adverse Events and Clinical Outcomes Among Patients With Cancer Receiving Immune Checkpoint Inhibitors. Cancer Control. 2022 Nov;29:10732748221130576. https://doi.org/10.1177/10732748221130576
[22] Shimozaki K, Sukawa Y, Sato Y, Horie S, Chida A, Tsugaru K, et al. Analysis of Risk Factors for Immune-Related Adverse Events in Various Solid Tumors Using Real-World Data. Future Oncol. 2021 Jul;17(20):2593–603. https://doi.org/10.2217/fon-2020-0861
[23] Ozawa R, Sonehara K, Hachiya T, Nozawa S, Agatsuma T, Yamamoto H, et al. Evaluation of the association between predictive factors and the development of immune‐related adverse events and prognostic factors for chemoimmunotherapy in patients with non‐small cell lung cancer: A multicenter retrospective study. Cancer Medicine. 2024 Aug;13(15):e70080. https://doi.org/10.1002/cam4.70080
[24] Hara Y, Baba Y, Toihata T, Harada K, Ogawa K, Iwatsuki M, et al. Immune-related adverse events and prognosis in patients with upper gastrointestinal cancer treated with nivolumab. J Gastrointest Oncol. 2022 Dec;13(6):2779–88. https://doi.org/10.21037/jgo-22-281
[25] Greally M, Chou JF, Chatila WK, Margolis M, Capanu M, Hechtman JF, et al. Clinical and Molecular Predictors of Response to Immune Checkpoint Inhibitors in Patients with Advanced Esophagogastric Cancer. Clinical Cancer Research. 2019 Oct 15;25(20):6160–9. https://doi.org/10.1158/1078-0432.CCR-18-3603
[26] Sato S, Oshima Y, Matsumoto Y, Seto Y, Yamashita H, Hayano K, et al. The new prognostic score for unresectable or recurrent gastric cancer treated with nivolumab: A multi‐institutional cohort study. Annals of Gastroent Surgery. 2021 Nov;5(6):794–803. https://doi.org/10.1002/ags3.12489
[27] Choe WH, Baik SK. Prostaglandin E2‐mediated immunosuppression and the role of albumin as its modulator. Hepatology. 2015 Mar;61(3):1080–2. https://doi.org/10.1002/hep.27644
[28] Ren W, Liang H, Sun J, Cheng Z, Liu W, Wu Y, et al. TNFAIP2 promotes HIF1α transcription and breast cancer angiogenesis by activating the Rac1-ERK-AP1 signaling axis. Cell Death Dis. 2024 Nov 13;15(11):821. https://doi.org/10.1038/s41419-024-07223-2
[29] Wang J, Al-Lamki RS. Tumor Necrosis Factor Receptor 2: Its Contribution to Acute Cellular Rejection and Clear Cell Renal Carcinoma. BioMed Research International. 2013;2013:1–11. https://doi.org/10.1155/2013/821310
[30] Langer HF. Platelets and Immune Responses During Thromboinflammation. Frontiers in Immunology 2019;10. https://doi.org/10.3389/fimmu.2019.0173
[31] Wojtukiewicz MZ, Sierko E, Hempel D, Tucker SC, Honn KV. Platelets and cancer angiogenesis nexus. Cancer Metastasis Rev. 2017 Jun;36(2):249–62. https://doi.org/10.1007/s10555-017-9673-1
[32] Jiang L, Luan Y, Miao X, Sun C, Li K, Huang Z, et al. Platelet releasate promotes breast cancer growth and angiogenesis via VEGF–integrin cooperative signalling. Br J Cancer. 2017 Aug;117(5):695–703. https://doi.org/10.1038/bjc.2017.214
[33] Haemmerle M, Stone RL, Menter DG, Afshar-Kharghan V, Sood AK. The Platelet Lifeline to Cancer: Challenges and Opportunities. Cancer Cell. 2018 Jun;33(6):965–83. https://doi.org/10.1016/j.ccell.2018.03.002
[34] Koupenova M, Clancy L, Corkrey HA, Freedman JE. Circulating Platelets as Mediators of Immunity, Inflammation, and Thrombosis. Circulation Research. 2018 Jan 19;122(2):337–51. https://doi.org/10.1161/CIRCRESAHA.117.310795
Type
Published
Data Availability Statement
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







