Artificial Intelligence–Enabled Dissection of Regulatory T Cellsin the Tumor Immune Microenvironment:From Mechanistic Insight to Population-Scale Inference
DOI:
https://doi.org/10.71321/s2yhxp11Keywords:
Artificial intelligence, Regulatory T cells, Tumor immune microenvironment, Immunotherapy, Precision oncologyAbstract
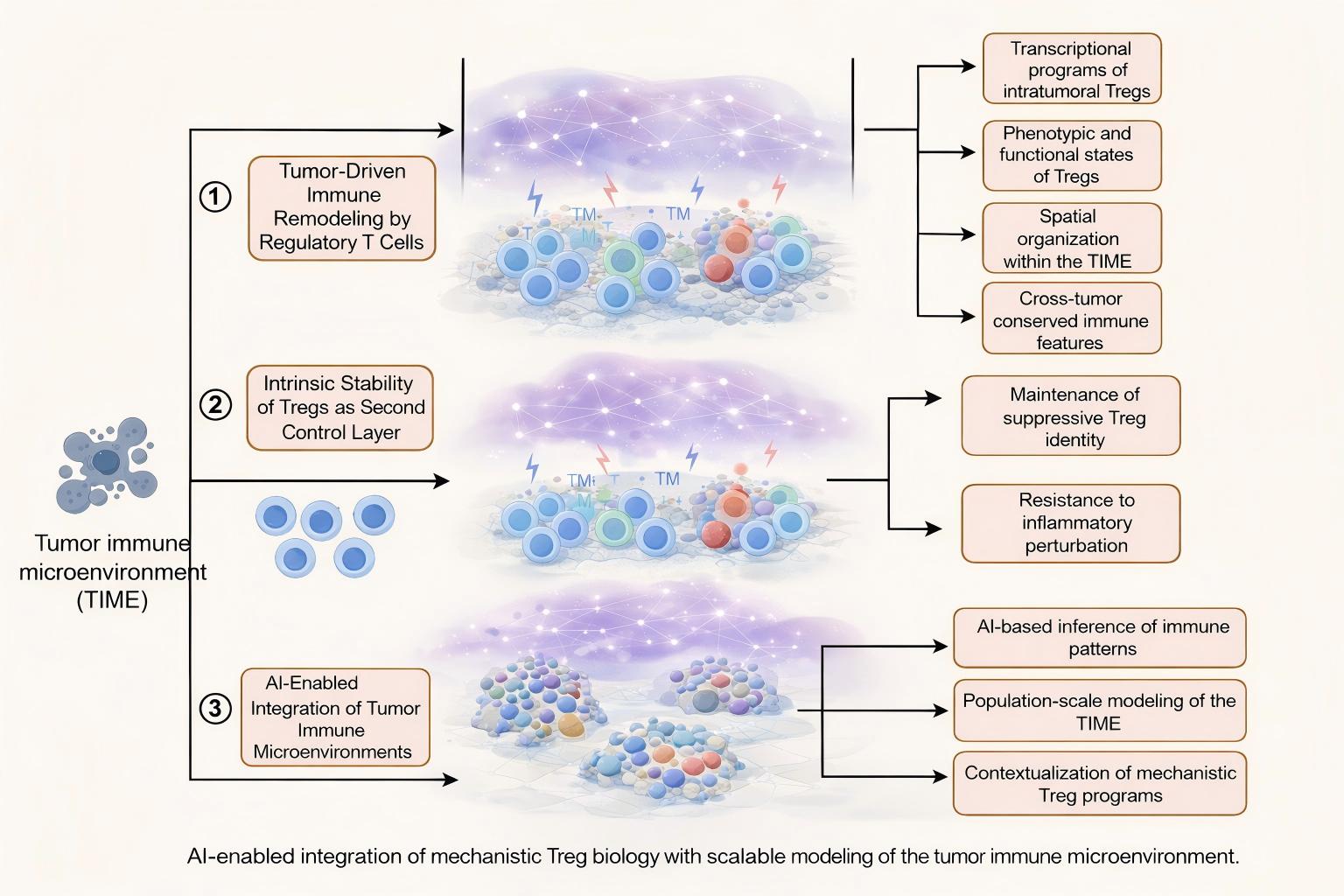
Background: Regulatory T cells (Tregs) are central regulators of immune tolerance in the tumor immune microenvironment (TIME) and are increasingly implicated in tumor immune evasion and immunotherapy resistance. While mechanistic studies have delineated pathways governing Treg recruitment and functional stability, extending these insights to spatial organization and population-level heterogeneity remains a major challenge.
Methods: In this correspondence, we integrate recent advances in tumor immunology with emerging artificial intelligence (AI)–based analytical frameworks to highlight how Treg-driven immune programs can be interrogated at scale. We draw on representative experimental mechanisms and AI-enabled multimodal modeling approaches, including virtual tumor microenvironment reconstruction and vision–language foundation models, to illustrate a mechanism-informed computational perspective.
Results: Evidence supports a layered model of tumor-driven Treg regulation, combining chemokine-mediated recruitment with intrinsic transcriptional stabilization within the TIME. AI-enabled approaches enable population-scale inference of Treg abundance, spatial distribution, and functional states, revealing clinically relevant heterogeneity and associations with differential immunotherapy responses that are difficult to capture using conventional experimental strategies alone.
Conclusion: The convergence of mechanistic Treg biology and AI-driven TIME modeling offers a conceptual framework for bridging experimental insight with real-world tumor heterogeneity. Mechanism-informed AI has the potential to refine immune stratification and guide Treg-targeted therapeutic strategies, highlighting a translational path forward for precision immuno-oncology.
References
[1] Li Z-C, Qin W, Liang D, & Zheng H. (2025). Biomedical AI: Evolving from digital to physical and biological intelligence. The Innovation Informatics, 1(1), 100010. https://doi.org/10.59717/j.xinn-inform.2025.100010
[2] Song Y, Jiang S, Peng Y, Qin C, Du Y, & Xu T. (2024). Effect of FGFR alteration on prognosis in 1963 urothelial carcinoma patients with immune checkpoint inhibitors: Implying combination of FGFR inhibitor and immunotherapy for FGFR-altered urothelial carcinoma. Pharmacol Res, 205, 107230. https://doi.org/10.1016/j.phrs.2024.107230
[3] Chen H, Li W, Hu W, Liu J, Zhang C, Wang Y, et al. (2025). Discovery of a novel tetrapeptide as glucose homeostasis modulator with bifunctionalities of targeting DPP-IV and microbiota. Imeta, 4(5), e70072. https://doi.org/10.1002/imt2.70072
[4] Luo Y, Li Y, Fang M, Wang S, Shao L, Zou R, et al. (2025). Multi-omics synergy in oncology: Unraveling the complex interplay of radiomic, genoproteomic, and pathological data. Intelligent Oncology, 1(1), 17-30. https://doi.org/https://doi.org/10.1016/j.intonc.2024.10.003
[5] Hao X, Liu L, Li S, Wang Q, Xu Z, & Liu X. (2025). The regulatory role and mechanism of TGFBI on Tregs in the immune microenvironment of clear cell renal cell carcinoma. Int J Surg, 111(12), 9134-9146. https://doi.org/10.1097/js9.0000000000003225
[6] Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, et al. (2004). Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med, 10(9), 942-949. https://doi.org/10.1038/nm1093
[7] Simpson TR, Li F, Montalvo-Ortiz W, Sepulveda MA, Bergerhoff K, Arce F, et al. (2013). Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J Exp Med, 210(9), 1695-1710. https://doi.org/10.1084/jem.20130579
[8] Glasner A, Rose SA, Sharma R, Gudjonson H, Chu T, Green JA, et al. (2023). Conserved transcriptional connectivity of regulatory T cells in the tumor microenvironment informs new combination cancer therapy strategies. Nat Immunol, 24(6), 1020-1035. https://doi.org/10.1038/s41590-023-01504-2
[9] Plitas G, Konopacki C, Wu K, Bos PD, Morrow M, Putintseva EV, et al. (2016). Regulatory T Cells Exhibit Distinct Features in Human Breast Cancer. Immunity, 45(5), 1122-1134. https://doi.org/10.1016/j.immuni.2016.10.032
[10] Ichiyama K, Long J, Kobayashi Y, Horita Y, Kinoshita T, Nakamura Y, et al. (2024). Transcription factor Ikzf1 associates with Foxp3 to repress gene expression in Treg cells and limit autoimmunity and anti-tumor immunity. Immunity, 57(9), 2043-2060.e2010. https://doi.org/10.1016/j.immuni.2024.07.010
[11] Valanarasu JMJ, Xu H, Usuyama N, Kim C, Wong C, Argaw P, et al. (2025). Multimodal AI generates virtual population for tumor microenvironment modeling. Cell, 10.1016/j.cell.2025.11.016. https://doi.org/10.1016/j.cell.2025.11.016
[12] Xiang J, Wang X, Zhang X, Xi Y, Eweje F, Chen Y, et al. (2025). A vision-language foundation model for precision oncology. Nature, 638(8051), 769-778. https://doi.org/10.1038/s41586-024-08378-w
Type
Published
Data Availability Statement
Not Applicable.
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







