Deep Brain Stimulation for Parkinson’s Disease: A Comprehensive Review of Efficacy, Influencing Factors, and Future Directions
DOI:
https://doi.org/10.71321/qwdy0v52Keywords:
Parkinson’s Disease, Movement disorders, Deep brain stimulation, ReviewAbstract
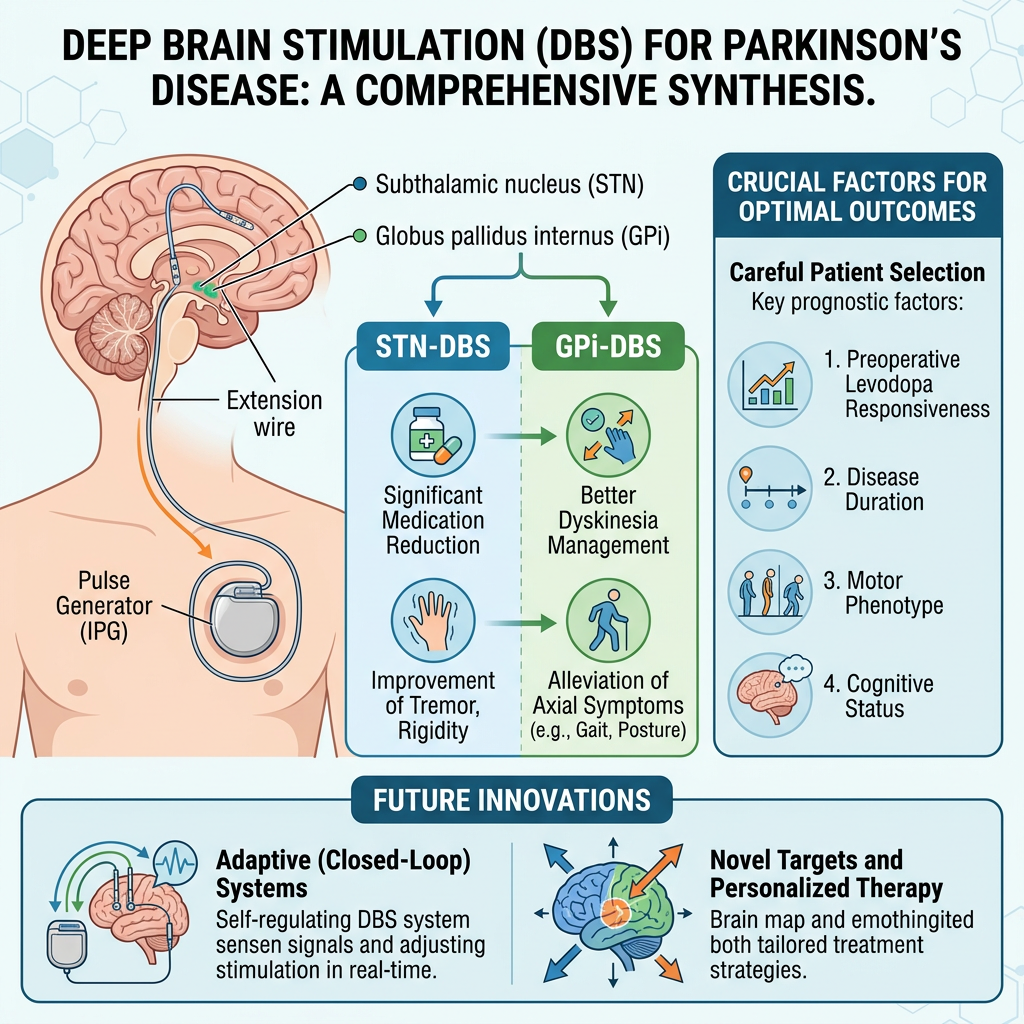
Deep Brain Stimulation (DBS) is a well-established surgical therapy for advanced Parkinson’s disease (PD), effectively alleviating motor symptoms and complications associated with long-term levodopa treatment. This review provides a comprehensive synthesis of current evidence, analyzing the efficacy of the two primary targets, the subthalamic nucleus (STN) and the globus pallidus internus (GPi), on both motor and non-motor symptoms. While STN-DBS allows significant medication reduction and GPi-DBS may better manage dyskinesias and axial symptoms, outcomes are highly dependent on careful patient selection. Key prognostic factors include preoperative levodopa responsiveness, disease duration, motor phenotype, and cognitive status. The review also critically evaluates technological advancements, such as adaptive closed-loop systems and novel targets, which aim to personalize therapy and address treatment-resistant symptoms. Ultimately, DBS represents a transformative intervention whose success hinges on integrated clinical decision-making and continued innovation.
References
[1] Wang S, Jiang Y, Yang A, Meng F, Zhang J. The Expanding Burden of Neurodegenerative Diseases: An Unmet Medical and Social Need. Aging Dis. 2024;16(5):2937–52. https://doi.org/10.14336/ad.2024.1071
[2] Bloem BR, Okun MS, Klein C. Parkinson's disease. Lancet. 2021;397(10291):2284–303. [PMID: 33848468 doi: 10.1016/s0140-6736(21)00218-x]
[3] Tolosa E, Garrido A, Scholz SW, Poewe W. Challenges in the diagnosis of Parkinson's disease. Lancet Neurol. 2021;20(5):385–97. https://doi.org/10.1016/s1474-4422(21)00030-2
[4] Trist BG, Hare DJ, Double KL. Oxidative stress in the aging substantia nigra and the etiology of Parkinson's disease. Aging Cell. 2019;18(6):e13031. https://doi.org/10.1111/acel.13031
[5] Schweitzer JS, Song B, Herrington TM, Park TY, Lee N, Ko S, et al. Personalized iPSC-Derived Dopamine Progenitor Cells for Parkinson's Disease. N Engl J Med. 2020;382(20):1926–32. https://doi.org/10.1056/NEJMoa1915872
[6] Morris HR, Spillantini MG, Sue CM, Williams-Gray CH. The pathogenesis of Parkinson's disease. Lancet. 2024;403(10423):293–304. https://doi.org/10.1016/s0140-6736(23)01478-2
[7] Hayes MT. Parkinson's Disease and Parkinsonism. Am J Med. 2019;132(7):802–7. https://doi.org/10.1016/j.amjmed.2019.03.001
[8] Tanner CM, Ostrem JL. Parkinson's Disease. N Engl J Med. 2024;391(5):442–52. https://doi.org/10.1056/NEJMra2401857
[9] Foltynie T, Bruno V, Fox S, Kühn AA, Lindop F, Lees AJ. Medical, surgical, and physical treatments for Parkinson's disease. Lancet. 2024;403(10423):305–24. https://doi.org/10.1016/s0140-6736(23)01429-0
[10] Verschuur CVM, Suwijn SR, Boel JA, Post B, Bloem BR, van Hilten JJ, et al. Randomized Delayed-Start Trial of Levodopa in Parkinson's Disease. N Engl J Med. 2019;380(4):315–24. https://doi.org/10.1056/NEJMoa1809983
[11] Falletti M, Asci F, Zampogna A, Patera M, Pinola G, Centonze D, et al. Rigidity in Parkinson's Disease: The Objective Effect of Levodopa. Mov Disord. 2025;40(4):727–38. https://doi.org/10.1002/mds.30114
[12] Wang S, Jiang Y, Yang A, Meng F, Zhang J. Continuous subcutaneous levodopa-carbidopa infusion for Parkinson's disease. Lancet Neurol. 2024;23(9):856. https://doi.org/10.1016/s1474-4422(24)00273-4
[13] Turcano P, Mielke MM, Bower JH, Parisi JE, Cutsforth-Gregory JK, Ahlskog JE, et al. Levodopa-induced dyskinesia in Parkinson disease: A population-based cohort study. Neurology. 2018;91(24):e2238–e43. https://doi.org/10.1212/wnl.0000000000006643
[14] Meng F, Hu W, Wang S, Tam J, Gao Y, Zhu XL, et al. Utilization, surgical populations, centers, coverages, regional balance, and their influential factors of deep brain stimulation for Parkinson's disease: a large-scale multicenter cross-sectional study from 1997 to 2021. Int J Surg. 2023;109(11):3322–36. https://doi.org/10.1097/js9.0000000000000603
[15] Deuschl G, Antonini A, Costa J, Śmiłowska K, Berg D, Corvol JC, et al. European Academy of Neurology/Movement Disorder Society-European Section Guideline on the Treatment of Parkinson's Disease: I. Invasive Therapies. Mov Disord. 2022;37(7):1360–74. https://doi.org/10.1002/mds.29066
[16] Reese R, Koeglsperger T, Schrader C, Tönges L, Deuschl G, Kühn AA, et al. Invasive therapies for Parkinson's disease: an adapted excerpt from the guidelines of the German Society of Neurology. J Neurol. 2025;272(3):219. https://doi.org/10.1007/s00415-025-12915-6
[17] Hariz M, Blomstedt P. Deep brain stimulation for Parkinson's disease. J Intern Med. 2022;292(5):764–78. https://doi.org/10.1111/joim.13541
[18] Wang S, Wang X, Zhao M, Li T, Zhang C, Wang M, et al. Long-term efficacy and cognitive effects of voltage-based deep brain stimulation for drug-resistant essential tremor. Clin Neurol Neurosurg. 2020;194:105940. https://doi.org/10.1016/j.clineuro.2020.105940
[19] Wang S, Zhao M, Li T, Zhang C, Zhou J, Wang M, et al. Long-term efficacy and cognitive effects of bilateral hippocampal deep brain stimulation in patients with drug-resistant temporal lobe epilepsy. Neurol Sci. 2021;42(1):225–33. https://doi.org/10.1007/s10072-020-04554-8
[20] Starr PA, Shivacharan RS, Goldberg E, Tröster AI, House PA, Giroux ML, et al. Five-Year Outcomes from Deep Brain Stimulation of the Subthalamic Nucleus for Parkinson Disease. JAMA Neurol. 2025;82(11):1181–90. https://doi.org/10.1001/jamaneurol.2025.3373
[21] Limousin P, Foltynie T. Long-term outcomes of deep brain stimulation in Parkinson disease. Nat Rev Neurol. 2019;15(4):234–42. https://doi.org/10.1038/s41582-019-0145-9
[22] Rughani A, Schwalb JM, Sidiropoulos C, Pilitsis J, Ramirez-Zamora A, Sweet JA, et al. Congress of Neurological Surgeons Systematic Review and Evidence-Based Guideline on Subthalamic Nucleus and Globus Pallidus Internus Deep Brain Stimulation for the Treatment of Patients With Parkinson's Disease: Executive Summary. Neurosurgery. 2018;82(6):753–6. https://doi.org/10.1093/neuros/nyy037
[23] Lachenmayer ML, Mürset M, Antih N, Debove I, Muellner J, Bompart M, et al. Subthalamic and pallidal deep brain stimulation for Parkinson's disease-meta-analysis of outcomes. NPJ Parkinsons Dis. 2021;7(1):77. https://doi.org/10.1038/s41531-021-00223-5
[24] de Bie RMA, Katzenschlager R, Swinnen B, Peball M, Lim SY, Mestre TA, et al. Update on Treatments for Parkinson's Disease Motor Fluctuations - An International Parkinson and Movement Disorder Society Evidence-Based Medicine Review. Mov Disord. 2025;40(5):776–94. https://doi.org/10.1002/mds.30162
[25] Deuschl G, Schade-Brittinger C, Krack P, Volkmann J, Schäfer H, Bötzel K, et al. A randomized trial of deep-brain stimulation for Parkinson's disease. N Engl J Med. 2006;355(9):896–908. https://doi.org/10.1056/NEJMoa060281
[26] Wong JK, Cauraugh JH, Ho KWD, Broderick M, Ramirez-Zamora A, Almeida L, et al. STN vs. GPi deep brain stimulation for tremor suppression in Parkinson disease: A systematic review and meta-analysis. Parkinsonism Relat Disord. 2019;58:56–62. https://doi.org/10.1016/j.parkreldis.2018.08.017
[27] Mostofi A, Evans JM, Partington-Smith L, Yu K, Chen C, Silverdale MA. Outcomes from deep brain stimulation targeting subthalamic nucleus and caudal zona incerta for Parkinson's disease. NPJ Parkinsons Dis. 2019;5:17. https://doi.org/10.1038/s41531-019-0089-1
[28] Dirkx MF, Bologna M. The pathophysiology of Parkinson's disease tremor. J Neurol Sci. 2022;435:120196. https://doi.org/10.1016/j.jns.2022.120196
[29] Herrington TM, Cheng JJ, Eskandar EN. Mechanisms of deep brain stimulation. J Neurophysiol. 2016;115(1):19–38. https://doi.org/10.1152/jn.00281.2015
[30] Deuter D, Mederer T, Kohl Z, Forras P, Rosengarth K, Schlabeck M, et al. Amelioration of Parkinsonian tremor evoked by DBS: which role play cerebello-(sub)thalamic fiber tracts? J Neurol. 2024;271(3):1451–61. https://doi.org/10.1007/s00415-023-12095-1
[31] Baumgartner AJ, Thompson JA, Kern DS, Ojemann SG. Novel targets in deep brain stimulation for movement disorders. Neurosurg Rev. 2022;45(4):2593–613. https://doi.org/10.1007/s10143-022-01770-y
[32] Azghadi A, Rajagopal MM, Atkinson KA, Holloway KL. Utility of GPI+VIM dual-lead deep brain stimulation for Parkinson's disease patients with significant residual tremor on medication. J Neurosurg. 2022;136(5):1364–70. https://doi.org/10.3171/2021.4.Jns21502
[33] Parihar R, Alterman R, Papavassiliou E, Tarsy D, Shih LC. Comparison of VIM and STN DBS for Parkinsonian Resting and Postural/Action Tremor. Tremor Other Hyperkinet Mov (N Y). 2015;5:321. https://doi.org/10.7916/d81v5d35
[34] Reich SG, Savitt JM. Parkinson's Disease. Med Clin North Am. 2019;103(2):337–50. https://doi.org/10.1016/j.mcna.2018.10.014
[35] Zampogna A, Suppa A, Bove F, Cavallieri F, Castrioto A, Meoni S, et al. Disentangling Bradykinesia and Rigidity in Parkinson's Disease: Evidence from Short- and Long-Term Subthalamic Nucleus Deep Brain Stimulation. Ann Neurol. 2024;96(2):234–46. https://doi.org/10.1002/ana.26961
[36] Kinugawa K, Mano T, Fujimura S, Takatani T, Miyasaka T, Sugie K. Bradykinesia and rigidity modulated by functional connectivity between the primary motor cortex and globus pallidus in Parkinson's disease. J Neural Transm (Vienna). 2023;130(12):1537–45. https://doi.org/10.1007/s00702-023-02688-5
[37] McGregor MM, Nelson AB. Circuit Mechanisms of Parkinson's Disease. Neuron. 2019;101(6):1042–56. https://doi.org/10.1016/j.neuron.2019.03.004
[38] Ni Z, Kim SJ, Phielipp N, Ghosh S, Udupa K, Gunraj CA, et al. Pallidal deep brain stimulation modulates cortical excitability and plasticity. Ann Neurol. 2018;83(2):352–62. https://doi.org/10.1002/ana.25156
[39] Collomb-Clerc A, Welter ML. Effects of deep brain stimulation on balance and gait in patients with Parkinson's disease: A systematic neurophysiological review. Neurophysiol Clin. 2015;45(4-5):371–88. https://doi.org/10.1016/j.neucli.2015.07.001
[40] Gao C, Liu J, Tan Y, Chen S. Freezing of gait in Parkinson's disease: pathophysiology, risk factors and treatments. Transl Neurodegener. 2020;9:12. https://doi.org/10.1186/s40035-020-00191-5
[41] Schlenstedt C, Shalash A, Muthuraman M, Falk D, Witt K, Deuschl G. Effect of high-frequency subthalamic neurostimulation on gait and freezing of gait in Parkinson's disease: a systematic review and meta-analysis. Eur J Neurol. 2017;24(1):18–26. https://doi.org/10.1111/ene.13167
[42] Tsuboi T, Lemos Melo Lobo Jofili Lopes J, Patel B, Legacy J, Moore K, Eisinger RS, et al. Parkinson's disease motor subtypes and bilateral GPi deep brain stimulation: One-year outcomes. Parkinsonism Relat Disord. 2020;75:7–13. https://doi.org/10.1016/j.parkreldis.2020.05.004
[43] Allert N, Volkmann J, Dotse S, Hefter H, Sturm V, Freund HJ. Effects of bilateral pallidal or subthalamic stimulation on gait in advanced Parkinson's disease. Mov Disord. 2001;16(6):1076–85. https://doi.org/10.1002/mds.1222
[44] Thevathasan W, Debu B, Aziz T, Bloem BR, Blahak C, Butson C, et al. Pedunculopontine nucleus deep brain stimulation in Parkinson's disease: A clinical review. Mov Disord. 2018;33(1):10–20. https://doi.org/10.1002/mds.27098
[45] Conway ZJ, Silburn PA, Perera T, O'Maley K, Cole MH. Low-frequency STN-DBS provides acute gait improvements in Parkinson's disease: a double-blinded randomised cross-over feasibility trial. J Neuroeng Rehabil. 2021;18(1):125. https://doi.org/10.1186/s12984-021-00921-4
[46] Jia F, Wagle Shukla A, Hu W, Ma Y, Zhang J, Almeida L, et al. Variable frequency deep brain stimulation of subthalamic nucleus to improve freezing of gait in Parkinson's disease. Natl Sci Rev. 2024;11(6):nwae187. https://doi.org/10.1093/nsr/nwae187
[47] Jia F, Hu W, Zhang J, Wagle Shukla A, Almeida L, Meng FG, et al. Variable frequency stimulation of subthalamic nucleus in Parkinson's disease: Rationale and hypothesis. Parkinsonism Relat Disord. 2017;39:27–30. https://doi.org/10.1016/j.parkreldis.2017.03.015
[48] Krack P, Volkmann J, Tinkhauser G, Deuschl G. Deep Brain Stimulation in Movement Disorders: From Experimental Surgery to Evidence-Based Therapy. Mov Disord. 2019;34(12):1795–810. https://doi.org/10.1002/mds.27860
[49] Neumann WJ, Steiner LA, Milosevic L. Neurophysiological mechanisms of deep brain stimulation across spatiotemporal resolutions. Brain. 2023;146(11):4456–68. https://doi.org/10.1093/brain/awad239
[50] Chiken S, Nambu A. Mechanism of Deep Brain Stimulation: Inhibition, Excitation, or Disruption? Neuroscientist. 2016;22(3):313–22. https://doi.org/10.1177/1073858415581986
[51] Li J, Mei S, Jia X, Zhang Y. Evaluation of the Direct Effect of Bilateral Deep Brain Stimulation of the Subthalamic Nucleus on Levodopa-Induced On-Dyskinesia in Parkinson's Disease. Front Neurol. 2021;12:595741. https://doi.org/10.3389/fneur.2021.595741
[52] Nassery A, Palmese CA, Sarva H, Groves M, Miravite J, Kopell BH. Psychiatric and Cognitive Effects of Deep Brain Stimulation for Parkinson's Disease. Curr Neurol Neurosci Rep. 2016;16(10):87. https://doi.org/10.1007/s11910-016-0690-1
[53] Jost ST, Sauerbier A, Visser-Vandewalle V, Ashkan K, Silverdale M, Evans J, et al. A prospective, controlled study of non-motor effects of subthalamic stimulation in Parkinson's disease: results at the 36-month follow-up. J Neurol Neurosurg Psychiatry. 2020;91(7):687–94. https://doi.org/10.1136/jnnp-2019-322614
[54] Combs HL, Folley BS, Berry DT, Segerstrom SC, Han DY, Anderson-Mooney AJ, et al. Cognition and Depression Following Deep Brain Stimulation of the Subthalamic Nucleus and Globus Pallidus Pars Internus in Parkinson's Disease: A Meta-Analysis. Neuropsychol Rev. 2015;25(4):439–54. https://doi.org/10.1007/s11065-015-9302-0
[55] Chang B, Mei J, Ni C, Niu C. Functional Connectivity and Anxiety Improvement After Subthalamic Nucleus Deep Brain Stimulation in Parkinson's Disease. Clin Interv Aging. 2023;18:1437–45. https://doi.org/10.2147/cia.S422605
[56] Zoon TJC, van Rooijen G, Balm G, Bergfeld IO, Daams JG, Krack P, et al. Apathy Induced by Subthalamic Nucleus Deep Brain Stimulation in Parkinson's Disease: A Meta-Analysis. Mov Disord. 2021;36(2):317–26. https://doi.org/10.1002/mds.28390
[57] Zhang Q, Kim J, Lu Q. Neuroimaging Advances in Neuropsychiatric Symptoms Associated with Parkinson’s Disease. Brain Conflux. 2025;1(1):e126. https://doi.org/10.71321/z4638252
[58] Bucur M, Papagno C. Deep Brain Stimulation in Parkinson Disease: A Meta-analysis of the Long-term Neuropsychological Outcomes. Neuropsychol Rev. 2023;33(2):307–46. https://doi.org/10.1007/s11065-022-09540-9
[59] Smeding HM, Speelman JD, Huizenga HM, Schuurman PR, Schmand B. Predictors of cognitive and psychosocial outcome after STN DBS in Parkinson's Disease. J Neurol Neurosurg Psychiatry. 2011;82(7):754–60. https://doi.org/10.1136/jnnp.2007.140012
[60] Howard CW, Reich M, Luo L, Pacheco-Barrios N, Alterman R, Rios AS, et al. Cognitive outcomes of deep brain stimulation depend on age and hippocampal connectivity in Parkinson's and Alzheimer's disease. Alzheimers Dement. 2025;21(8):e70498. https://doi.org/10.1002/alz.70498
[61] Luo B, Zou Y, Yan J, Sun J, Wei X, Chang L, et al. Altered Cognitive Networks Connectivity in Parkinson's Disease During the Microlesion Period After Deep Brain Stimulation. CNS Neurosci Ther. 2024;30(12):e70184. https://doi.org/10.1111/cns.70184
[62] Devignes Q, Daoudi S, Viard R, Lopes R, Betrouni N, Kuchcinski G, et al. Heterogeneity of PD-MCI in Candidates to Subthalamic Deep Brain Stimulation: Associated Cortical and Subcortical Modifications. J Parkinsons Dis. 2022;12(5):1507–26. https://doi.org/10.3233/jpd-223232
[63] Kim HJ, Jeon BS, Paek SH, Lee KM, Kim JY, Lee JY, et al. Long-term cognitive outcome of bilateral subthalamic deep brain stimulation in Parkinson's disease. J Neurol. 2014;261(6):1090–6. https://doi.org/10.1007/s00415-014-7321-z
[64] Zuzuárregui JRP, Ostrem JL. The Impact of Deep Brain Stimulation on Sleep in Parkinson's Disease: An update. J Parkinsons Dis. 2020;10(2):393–404. https://doi.org/10.3233/jpd-191862
[65] Choi JH, Kim HJ, Lee JY, Yoo D, Im JH, Paek SH, et al. Long-term effects of bilateral subthalamic nucleus stimulation on sleep in patients with Parkinson's disease. PLoS One. 2019;14(8):e0221219. https://doi.org/10.1371/journal.pone.0221219
[66] Casoni F, Galbiati TF, Ferini-Strambi L, Marelli S, Zucconi M, Servello D. DBS in restless legs syndrome: a new therapeutic approach? Sleep Med. 2020;76:155–7. https://doi.org/10.1016/j.sleep.2020.10.022
[67] Zibetti M, Rizzi L, Colloca L, Cinquepalmi A, Angrisano S, Castelli L, et al. Probable REM sleep behaviour disorder and STN-DBS outcome in Parkinson's Disease. Parkinsonism Relat Disord. 2010;16(4):265–9. https://doi.org/10.1016/j.parkreldis.2010.01.001
[68] Besse-Pinot E, Pereira B, Durif F, Fantini ML, Durand E, Debilly B, et al. Preoperative REM Sleep Behavior Disorder and Subthalamic Nucleus Deep Brain Stimulation Outcome in Parkinson Disease 1 Year After Surgery. Neurology. 2021;97(20):e1994–e2006. https://doi.org/10.1212/wnl.0000000000012862
[69] Chahine LM, Ahmed A, Sun Z. Effects of STN DBS for Parkinson's disease on restless legs syndrome and other sleep-related measures. Parkinsonism Relat Disord. 2011;17(3):208–11. https://doi.org/10.1016/j.parkreldis.2010.11.017
[70] Dulski J, Wąż P, Konkel A, Grabowski K, Libionka W, Schinwelski M, et al. The Impact of Subthalamic Deep Brain Stimulation on Restless Legs Syndrome in Parkinson's Disease. Neuromodulation. 2022;25(6):904–10. https://doi.org/10.1111/ner.13462
[71] Wang S, Hu W, Gao Y, Wang A, Chen L, Liang Z, et al. Deep brain stimulation surgical timing, outcomes, and prognostic factors in patients with Parkinson's disease: A Chinese retrospective multicenter cohort study. PLoS Med. 2025;22(8):e1004670. https://doi.org/10.1371/journal.pmed.1004670
[72] Cavallieri F, Fraix V, Bove F, Mulas D, Tondelli M, Castrioto A, et al. Predictors of Long-Term Outcome of Subthalamic Stimulation in Parkinson Disease. Ann Neurol. 2021;89(3):587–97. https://doi.org/10.1002/ana.25994
[73] Piboolnurak P, Lang AE, Lozano AM, Miyasaki JM, Saint-Cyr JA, Poon YY, et al. Levodopa response in long-term bilateral subthalamic stimulation for Parkinson's disease. Mov Disord. 2007;22(7):990–7. https://doi.org/10.1002/mds.21482
[74] Fabbri M, Zibetti M, Rizzone MG, Giannini G, Borellini L, Stefani A, et al. Should We Consider Deep Brain Stimulation Discontinuation in Late-Stage Parkinson's Disease? Mov Disord. 2020;35(8):1379–87. https://doi.org/10.1002/mds.28091
[75] Zheng Y, Wei L, Ang SYL, Ng HL, Au WL, Tay KY, et al. Outcomes after deep brain stimulation for elderly versus non-elderly patients with Parkinson's disease. Clin Neurol Neurosurg. 2024;242:108319. https://doi.org/10.1016/j.clineuro.2024.108319
[76] Schuepbach WM, Rau J, Knudsen K, Volkmann J, Krack P, Timmermann L, et al. Neurostimulation for Parkinson's disease with early motor complications. N Engl J Med. 2013;368(7):610–22. https://doi.org/10.1056/NEJMoa1205158
[77] Lee SJ, Kim JS, Lee KS, An JY, Kim W, Kim YI, et al. The severity of leukoaraiosis correlates with the clinical phenotype of Parkinson's disease. Arch Gerontol Geriatr. 2009;49(2):255–9. https://doi.org/10.1016/j.archger.2008.09.005
[78] Rundek T, Shpiner DS, Margolesky J. Perivascular spaces in basal ganglia - An innocent bystander in Parkinson's disease? Mov Disord. 2019;34(11):1585–7. https://doi.org/10.1002/mds.27824
[79] Katz M, Luciano MS, Carlson K, Luo P, Marks WJ, Jr., Larson PS, et al. Differential effects of deep brain stimulation target on motor subtypes in Parkinson's disease. Ann Neurol. 2015;77(4):710–9. https://doi.org/10.1002/ana.24374
[80] Stebbins GT, Goetz CG, Burn DJ, Jankovic J, Khoo TK, Tilley BC. How to identify tremor dominant and postural instability/gait difficulty groups with the movement disorder society unified Parkinson's disease rating scale: comparison with the unified Parkinson's disease rating scale. Mov Disord. 2013;28(5):668–70. https://doi.org/10.1002/mds.25383
[81] Nutt JG. Motor subtype in Parkinson's disease: Different disorders or different stages of disease? Mov Disord. 2016;31(7):957–61. https://doi.org/10.1002/mds.26657
[82] Krauss JK, Lipsman N, Aziz T, Boutet A, Brown P, Chang JW, et al. Technology of deep brain stimulation: current status and future directions. Nat Rev Neurol. 2021;17(2):75–87. https://doi.org/10.1038/s41582-020-00426-z
[83] Lozano AM, Lipsman N, Bergman H, Brown P, Chabardes S, Chang JW, et al. Deep brain stimulation: current challenges and future directions. Nat Rev Neurol. 2019;15(3):148–60. https://doi.org/10.1038/s41582-018-0128-2
[84] Wang S, Zhu G, Shi L, Zhang C, Wu B, Yang A, et al. Closed-Loop Adaptive Deep Brain Stimulation in Parkinson's Disease: Procedures to Achieve It and Future Perspectives. J Parkinsons Dis. 2023;13(4):453–71. https://doi.org/10.3233/jpd-225053
[85] Neumann WJ, Gilron R, Little S, Tinkhauser G. Adaptive Deep Brain Stimulation: From Experimental Evidence Toward Practical Implementation. Mov Disord. 2023;38(6):937–48. https://doi.org/10.1002/mds.29415
[86] Little S, Pogosyan A, Neal S, Zavala B, Zrinzo L, Hariz M, et al. Adaptive deep brain stimulation in advanced Parkinson disease. Ann Neurol. 2013;74(3):449–57. https://doi.org/10.1002/ana.23951
[87] Little S, Brown P. Debugging Adaptive Deep Brain Stimulation for Parkinson's Disease. Mov Disord. 2020;35(4):555–61. https://doi.org/10.1002/mds.27996
[88] Bronte-Stewart HM, Beudel M, Ostrem JL, Little S, Almeida L, Ramirez-Zamora A, et al. Long-Term Personalized Adaptive Deep Brain Stimulation in Parkinson Disease: A Nonrandomized Clinical Trial. JAMA Neurol. 2025;82(11):1171–80. https://doi.org/10.1001/jamaneurol.2025.2781
[89] Oehrn CR, Cernera S, Hammer LH, Shcherbakova M, Yao J, Hahn A, et al. Chronic adaptive deep brain stimulation versus conventional stimulation in Parkinson's disease: a blinded randomized feasibility trial. Nat Med. 2024;30(11):3345–56. https://doi.org/10.1038/s41591-024-03196-z
[90] Steigerwald F, Matthies C, Volkmann J. Directional Deep Brain Stimulation. Neurotherapeutics. 2019;16(1):100–4. https://doi.org/10.1007/s13311-018-0667-7
[91] Cherif S, Tempier N, Yeche M, Temiz G, Perrière J, Romanato M, et al. Directional Subthalamic Deep Brain Stimulation Better Improves Gait and Balance Disorders in Parkinson's Disease Patients: A Randomized Controlled Study. Ann Neurol. 2024;97(1):149–62. https://doi.org/10.1002/ana.27099
[92] Dembek TA, Reker P, Visser-Vandewalle V, Wirths J, Treuer H, Klehr M, et al. Directional DBS increases side-effect thresholds-A prospective, double-blind trial. Mov Disord. 2017;32(10):1380–8. https://doi.org/10.1002/mds.27093
[93] Lin F, Wu D, Lin C, Cai H, Chen L, Cai G, et al. Pedunculopontine Nucleus Deep Brain Stimulation Improves Gait Disorder in Parkinson's Disease: A Systematic Review and Meta-analysis. Neurochem Res. 2020;45(4):709–19. https://doi.org/10.1007/s11064-020-02962-y
Type
Published
Data Availability Statement
The data that support the findings of this study are included in the article/supplementary material.
Issue
Section
License
Copyright (c) 2026 Brain Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







