Regulatory T Cells in Precision Immunotherapy: Mechanistic Insights and Translational Advances of Low-dose Interleukin-2
DOI:
https://doi.org/10.71321/xk8wzt47Keywords:
Regulatory T Cells, Low-dose Interleukin-2, Foxp3, Immune tolerance, Precision immunotherapy, Translational medicineAbstract
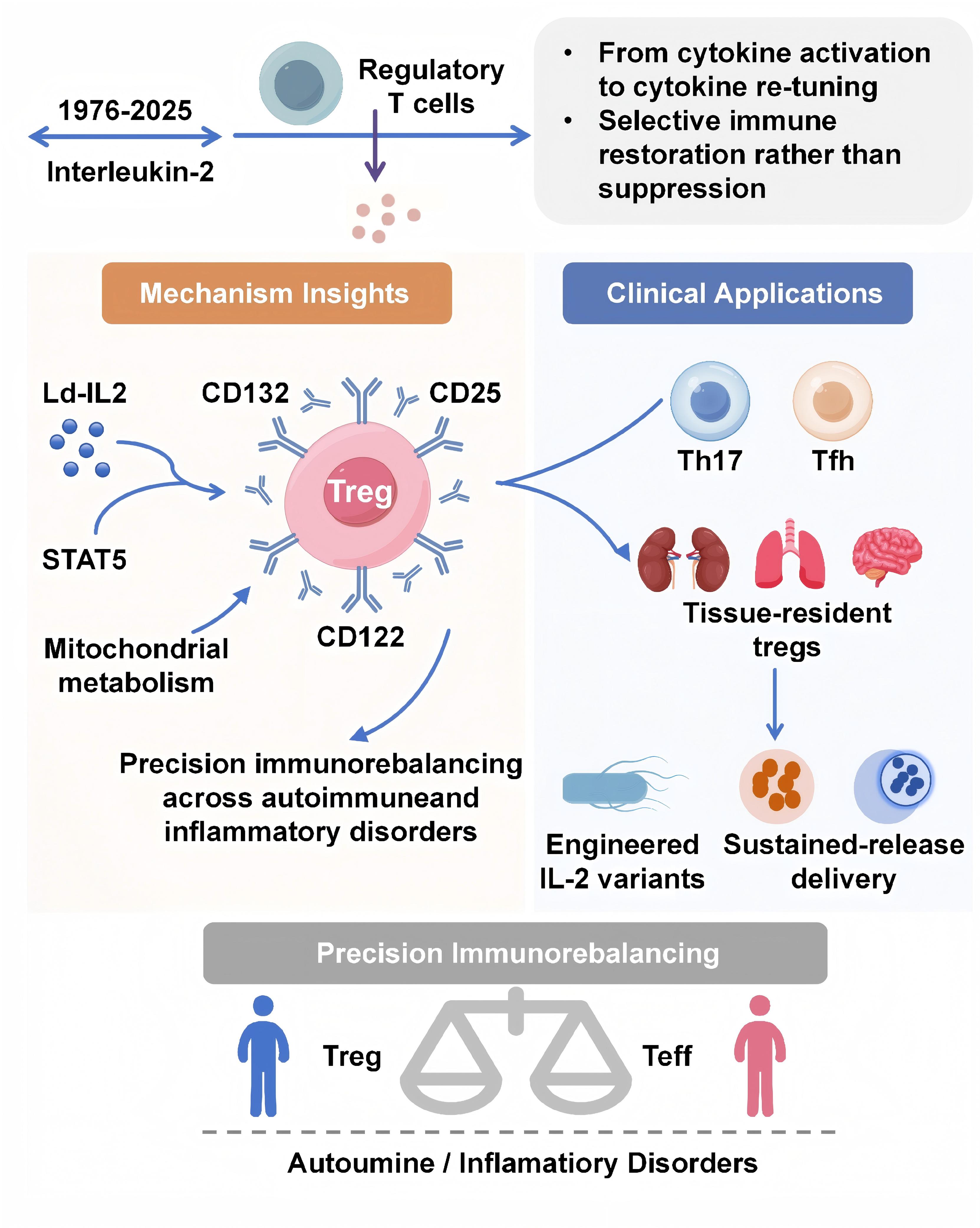
Regulatory T cells (Tregs) are the central guardians of immune tolerance, safeguarding against autoimmune and inflammatory damage through Foxp3-dependent transcriptional programs. Recent breakthroughs in precision immunotherapy have revived interest in low-dose interleukin-2 (Ld-IL-2), a cytokine-based strategy that selectively expands and activates Tregs via the high-affinity IL-2 receptor (CD25). This review summarizes emerging mechanistic insights into how Ld-IL-2 orchestrates multilevel immune rebalancing and highlights its translational progress from molecular engineering to clinical applications across autoimmune diseases. We integrated recent findings from cellular, metabolic, and systems immunology studies, together with our own multi-center clinical trial data, to outline the dynamic networks linking IL-2 signaling, Treg plasticity, and immune homeostasis. Ld-IL-2 exerts a dose-dependent biphasic effect on the immune system, selectively enhancing Treg survival and function while restraining pathogenic Th17, Tfh, and Teff subsets. Beyond classical STAT5-FOXP3 activation, recent studies reveal that IL-2 reprograms Treg metabolism toward oxidative phosphorylation, stabilizes Foxp3 epigenetic landscapes, and coordinates intercellular communication through exosomal and tissue-resident networks. Innovations in topologically engineered IL-2 variants and sustained-release delivery systems (e.g., polylactic-acid microsphere–exosome composites) further extend the precision and durability of Treg-directed therapy. Clinical evidence from SLE, Sjögren’s disease, and relapsing polychondritis confirms robust immune restoration and favorable safety profiles within defined dose windows. By selectively activating the Treg axis and reprogramming immune homeostasis, low-dose IL-2 represents a paradigm for precision immunotherapy. Integrating molecular engineering and targeted delivery strategies will enable next-generation cytokine therapies to achieve durable immune tolerance across autoimmune and inflammatory disorders.
References
[1] Morgan DA, Ruscetti FW, & Gallo R. (1976). Selective in vitro growth of T lymphocytes from normal human bone marrows. Science, 193(4257), 1007-1008. https://doi.org/10.1126/science.181845
[2] Rosenberg SA. (2014). IL-2: the first effective immunotherapy for human cancer. J Immunol, 192(12), 5451-5458. https://doi.org/10.4049/jimmunol.1490019
[3] Hu W, Dolsten GA, Wang EY, Beroshvili G, Wang ZM, Ghelani AP, et al. (2025). Temporal and context-dependent requirements for the transcription factor Foxp3 expression in regulatory T cells. Nat Immunol, 10.1038/s41590-025-02295-4. https://doi.org/10.1038/s41590-025-02295-4
[4] Boyman O, & Sprent J. (2012). The role of interleukin-2 during homeostasis and activation of the immune system. Nat Rev Immunol, 12(3), 180-190. https://doi.org/10.1038/nri3156
[5] Wang X, Rickert M, & Garcia KC. (2005). Structure of the quaternary complex of interleukin-2 with its alpha, beta, and gammac receptors. Science, 310(5751), 1159-1163. https://doi.org/10.1126/science.1117893
[6] Overwijk WW, Tagliaferri MA, & Zalevsky J. (2021). Engineering IL-2 to Give New Life to T Cell Immunotherapy. Annu Rev Med, 72, 281-311. https://doi.org/10.1146/annurev-med-073118-011031
[7] Koreth J, Matsuoka K, Kim HT, McDonough SM, Bindra B, Alyea EP, 3rd, et al. (2011). Interleukin-2 and regulatory T cells in graft-versus-host disease. N. Engl. J. Med., 365(22), 2055-2066. https://doi.org/10.1056/NEJMoa1108188
[8] He J, Zhang R, Shao M, Zhao X, Miao M, Chen J, et al. (2020). Efficacy and safety of low-dose IL-2 in the treatment of systemic lupus erythematosus: a randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis., 79(1), 141-149. https://doi.org/10.1136/annrheumdis-2019-215396
[9] He J, Chen J, Miao M, Zhang R, Cheng G, Wang Y, et al. (2022). Efficacy and Safety of Low-Dose Interleukin 2 for Primary Sjögren Syndrome: A Randomized Clinical Trial. JAMA Netw Open, 5(11), e2241451. https://doi.org/10.1001/jamanetworkopen.2022.41451
[10] Hartemann A, Bensimon G, Payan CA, Jacqueminet S, Bourron O, Nicolas N, et al. (2013). Low-dose interleukin 2 in patients with type 1 diabetes: a phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol, 1(4), 295-305. https://doi.org/10.1016/S2213-8587(13)70113-X
[11] Mitra S, & Leonard WJ. (2018). Biology of IL-2 and its therapeutic modulation: Mechanisms and strategies. J Leukoc Biol, 103(4), 643-655. https://doi.org/10.1002/jlb.2ri0717-278r
[12] Abbas AK, Trotta E, D RS, Marson A, & Bluestone JA. (2018). Revisiting IL-2: Biology and therapeutic prospects. Sci Immunol, 3(25). https://doi.org/10.1126/sciimmunol.aat1482
[13] Johnston JA, Kawamura M, Kirken RA, Chen YQ, Blake TB, Shibuya K, et al. (1994). Phosphorylation and activation of the Jak-3 Janus kinase in response to interleukin-2. Nature, 370(6485), 151-153. https://doi.org/10.1038/370151a0
[14] Ghoreschi K, Laurence A, & O'Shea JJ. (2009). Janus kinases in immune cell signaling. Immunol Rev, 228(1), 273-287. https://doi.org/10.1111/j.1600-065X.2008.00754.x
[15] Lin JX, Li P, Liu D, Jin HT, He J, Ata Ur Rasheed M, et al. (2012). Critical Role of STAT5 transcription factor tetramerization for cytokine responses and normal immune function. Immunity, 36(4), 586-599. https://doi.org/10.1016/j.immuni.2012.02.017
[16] Chinen T, Kannan AK, Levine AG, Fan X, Klein U, Zheng Y, et al. (2016). An essential role for the IL-2 receptor in T(reg) cell function. Nat Immunol, 17(11), 1322-1333. https://doi.org/10.1038/ni.3540
[17] Feng Y, van der Veeken J, Shugay M, Putintseva EV, Osmanbeyoglu HU, Dikiy S, et al. (2015). A mechanism for expansion of regulatory T-cell repertoire and its role in self-tolerance. Nature, 528(7580), 132-136. https://doi.org/10.1038/nature16141
[18] Ise W, Kohyama M, Nutsch KM, Lee HM, Suri A, Unanue ER, et al. (2010). CTLA-4 suppresses the pathogenicity of self antigen-specific T cells by cell-intrinsic and cell-extrinsic mechanisms. Nat Immunol, 11(2), 129-135. https://doi.org/10.1038/ni.1835
[19] Zheng Y, Josefowicz SZ, Kas A, Chu TT, Gavin MA, & Rudensky AY. (2007). Genome-wide analysis of Foxp3 target genes in developing and mature regulatory T cells. Nature, 445(7130), 936-940. https://doi.org/10.1038/nature05563
[20] Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, et al. (2008). CTLA-4 control over Foxp3+ regulatory T cell function. Science, 322(5899), 271-275. https://doi.org/10.1126/science.1160062
[21] Yao Z, Kanno Y, Kerenyi M, Stephens G, Durant L, Watford WT, et al. (2007). Nonredundant roles for Stat5a/b in directly regulating Foxp3. Blood, 109(10), 4368-4375. https://doi.org/10.1182/blood-2006-11-055756
[22] Feng Y, Arvey A, Chinen T, van der Veeken J, Gasteiger G, & Rudensky AY. (2014). Control of the inheritance of regulatory T cell identity by a cis element in the Foxp3 locus. Cell, 158(4), 749-763. https://doi.org/10.1016/j.cell.2014.07.031
[23] Shevyrev D, & Tereshchenko V. (2019). Treg Heterogeneity, Function, and Homeostasis. Front Immunol, 10, 3100. https://doi.org/10.3389/fimmu.2019.03100
[24] Wang X, Szymczak-Workman AL, Gravano DM, Workman CJ, Green DR, & Vignali DA. (2012). Preferential control of induced regulatory T cell homeostasis via a Bim/Bcl-2 axis. Cell Death Dis, 3(2), e270. https://doi.org/10.1038/cddis.2012.9
[25] Barahona de Brito C, & Patra AK. (2022). NFAT Factors Are Dispensable for the Development but Are Critical for the Maintenance of Foxp3(+) Regulatory T Cells. Cells, 11(9). https://doi.org/10.3390/cells11091397
[26] Wieczorek G, Asemissen A, Model F, Turbachova I, Floess S, Liebenberg V, et al. (2009). Quantitative DNA methylation analysis of FOXP3 as a new method for counting regulatory T cells in peripheral blood and solid tissue. Cancer Res, 69(2), 599-608. https://doi.org/10.1158/0008-5472.Can-08-2361
[27] Chen Q, Kim YC, Laurence A, Punkosdy GA, & Shevach EM. (2011). IL-2 controls the stability of Foxp3 expression in TGF-beta-induced Foxp3+ T cells in vivo. J Immunol, 186(11), 6329-6337. https://doi.org/10.4049/jimmunol.1100061
[28] Bending D, Pesenacker AM, Ursu S, Wu Q, Lom H, Thirugnanabalan B, et al. (2014). Hypomethylation at the regulatory T cell-specific demethylated region in CD25hi T cells is decoupled from FOXP3 expression at the inflamed site in childhood arthritis. J Immunol, 193(6), 2699-2708. https://doi.org/10.4049/jimmunol.1400599
[29] Huss DJ, Mehta DS, Sharma A, You X, Riester KA, Sheridan JP, et al. (2015). In vivo maintenance of human regulatory T cells during CD25 blockade. J Immunol, 194(1), 84-92. https://doi.org/10.4049/jimmunol.1402140
[30] Toker A, Engelbert D, Garg G, Polansky JK, Floess S, Miyao T, et al. (2013). Active demethylation of the Foxp3 locus leads to the generation of stable regulatory T cells within the thymus. J Immunol, 190(7), 3180-3188. https://doi.org/10.4049/jimmunol.1203473
[31] Correa LO, Jordan MS, & Carty SA. (2020). DNA Methylation in T-Cell Development and Differentiation. Crit Rev Immunol, 40(2), 135-156. https://doi.org/10.1615/CritRevImmunol.2020033728
[32] Chen C, Wang Z, Ding Y, Wang L, Wang S, Wang H, et al. (2022). DNA Methylation: From Cancer Biology to Clinical Perspectives. Front Biosci (Landmark Ed), 27(12), 326. https://doi.org/10.31083/j.fbl2712326
[33] Guo C, Liu Q, Zong D, Zhang W, Zuo Z, Yu Q, et al. (2022). Single-cell transcriptome profiling and chromatin accessibility reveal an exhausted regulatory CD4+ T cell subset in systemic lupus erythematosus. Cell Rep, 41(6), 111606. https://doi.org/10.1016/j.celrep.2022.111606
[34] Bonazzi S, d'Hennezel E, Beckwith REJ, Xu L, Fazal A, Magracheva A, et al. (2023). Discovery and characterization of a selective IKZF2 glue degrader for cancer immunotherapy. Cell Chem Biol, 30(3), 235-247.e212. https://doi.org/10.1016/j.chembiol.2023.02.005
[35] Xin Y, Yang M, Zhao Z, He Z, Mei Y, Xiong F, et al. (2025). AIM2 deficiency in CD4(+) T cells promotes psoriasis-like inflammation by regulating Th17-Treg axis via AIM2-IKZF2 pathway. J Autoimmun, 150, 103351. https://doi.org/10.1016/j.jaut.2024.103351
[36] McDonald-Hyman C, Aguilar EG, Compeer EB, Zaiken MC, Rhee SY, Mohamed FA, et al. (2025). Acetyl-CoA carboxylase-1 inhibition increases regulatory T-Cell metabolism and graft-vs-host disease treatment efficacy via mitochondrial fusion. J Clin Invest, 10.1172/jci182480. https://doi.org/10.1172/jci182480
[37] Han J, Zhou Z, Wang H, Chen Y, Li W, Dai M, et al. (2025). Dysfunctional glycolysis-UCP2-fatty acid oxidation promotes CTLA4(int)FOXP3(int) regulatory T-cell production in rheumatoid arthritis. Mol Med, 31(1), 310. https://doi.org/10.1186/s10020-025-01372-6
[38] Feng R, Xiao X, Wang Y, Huang B, Chen J, Cheng G, et al. (2024). Metabolic impact of low dose IL-2 therapy for primary Sjögren's Syndrome in a double-blind, randomized clinical trial. Clin Rheumatol, 43(12), 3789-3798. https://doi.org/10.1007/s10067-024-07165-2
[39] Duan W, Ding Y, Yu X, Ma D, Yang B, Li Y, et al. (2019). Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production. Am J Transl Res, 11(4), 2393-2402.
[40] Pokhrel RH, Kang B, Timilshina M, & Chang JH. (2022). AMPK Amplifies IL2-STAT5 Signaling to Maintain Stability of Regulatory T Cells in Aged Mice. Int J Mol Sci, 23(20). https://doi.org/10.3390/ijms232012384
[41] De La Rosa J, Geller AM, LeGros HL, Jr., & Kotb M. (1992). Induction of interleukin 2 production but not methionine adenosyltransferase activity or S-adenosylmethionine turnover in Jurkat T-cells. Cancer Res, 52(12), 3361-3366.
[42] Sahin E, & Sahin M. (2019). Epigenetical Targeting of the FOXP3 Gene by S-Adenosylmethionine Diminishes the Suppressive Capacity of Regulatory T Cells Ex Vivo and Alters the Expression Profiles. J Immunother, 42(1), 11-22. https://doi.org/10.1097/cji.0000000000000247
[43] Field CS, Baixauli F, Kyle RL, Puleston DJ, Cameron AM, Sanin DE, et al. (2020). Mitochondrial Integrity Regulated by Lipid Metabolism Is a Cell-Intrinsic Checkpoint for Treg Suppressive Function. Cell Metab, 31(2), 422-437.e425. https://doi.org/10.1016/j.cmet.2019.11.021
[44] Zhang X, Wang G, Bi Y, Jiang Z, & Wang X. (2022). Inhibition of glutaminolysis ameliorates lupus by regulating T and B cell subsets and downregulating the mTOR/P70S6K/4EBP1 and NLRP3/caspase-1/IL-1β pathways in MRL/lpr mice. Int Immunopharmacol, 112, 109133. https://doi.org/10.1016/j.intimp.2022.109133
[45] Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, et al. (2007). Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med, 204(6), 1257-1265. https://doi.org/10.1084/jem.20062512
[46] Arpaia N, Green JA, Moltedo B, Arvey A, Hemmers S, Yuan S, et al. (2015). A Distinct Function of Regulatory T Cells in Tissue Protection. Cell, 162(5), 1078-1089. https://doi.org/10.1016/j.cell.2015.08.021
[47] Zhang P, Wang J, Miao J, & Zhu P. (2025). The dual role of tissue regulatory T cells in tissue repair: return to homeostasis or fibrosis. Front Immunol, 16, 1560578. https://doi.org/10.3389/fimmu.2025.1560578
[48] Hilaire M, Mimoun A, Cagnet L, Villette R, Roubanis A, Sentenac H, et al. (2025). Neonatal regulatory T cells persist into adulthood across multiple tissues with high enrichment in the skin. Sci Adv, 11(40), eadx8037. https://doi.org/10.1126/sciadv.adx8037
[49] Fallegger A, Priola M, Artola-Borán M, Núñez NG, Wild S, Gurtner A, et al. (2022). TGF-β production by eosinophils drives the expansion of peripherally induced neuropilin(-) RORγt(+) regulatory T-cells during bacterial and allergen challenge. Mucosal Immunol, 15(3), 504-514. https://doi.org/10.1038/s41385-022-00484-0
[50] Zhou X, Tang J, Cao H, Fan H, & Li B. (2015). Tissue resident regulatory T cells: novel therapeutic targets for human disease. Cell Mol Immunol, 12(5), 543-552. https://doi.org/10.1038/cmi.2015.23
[51] Wang Y, Feng R, Cheng G, Huang B, Tian J, Gan Y, et al. (2022). Low Dose Interleukin-2 Ameliorates Sjögren's Syndrome in a Murine Model. Front Med (Lausanne), 9, 887354. https://doi.org/10.3389/fmed.2022.887354
[52] Xie F, Zhou X, Fang M, Li H, Su P, Tu Y, et al. (2019). Extracellular Vesicles in Cancer Immune Microenvironment and Cancer Immunotherapy. Adv Sci (Weinh), 6(24), 1901779. https://doi.org/10.1002/advs.201901779
[53] Xia Y, Jiang H, Wang C, Liu Z, Gao H, Yu JF, et al. (2025). Treg cell-derived exosomal miR-21 promotes osteogenic differentiation of periodontal ligament stem cells. BMC Oral Health, 25(1), 1465. https://doi.org/10.1186/s12903-025-06770-0
[54] Okoye IS, Coomes SM, Pelly VS, Czieso S, Papayannopoulos V, Tolmachova T, et al. (2014). MicroRNA-containing T-regulatory-cell-derived exosomes suppress pathogenic T helper 1 cells. Immunity, 41(1), 89-103. https://doi.org/10.1016/j.immuni.2014.05.019
[55] Rahmani M, Mohammadnia-Afrouzi M, Nouri HR, Fattahi S, Akhavan-Niaki H, & Mostafazadeh A. (2018). Human PBMCs fight or flight response to starvation stress: Increased T-reg, FOXP3, and TGF-β1 with decreased miR-21 and Constant miR-181c levels. Biomed Pharmacother, 108, 1404-1411. https://doi.org/10.1016/j.biopha.2018.09.163
[56] Gertel S, Polachek A, Elkayam O, & Furer V. (2022). Lymphocyte activation gene-3 (LAG-3) regulatory T cells: An evolving biomarker for treatment response in autoimmune diseases. Autoimmun Rev, 21(6), 103085. https://doi.org/10.1016/j.autrev.2022.103085
[57] Ravichandran R, Itabashi Y, Fleming T, Bansal S, Bowen S, Poulson C, et al. (2022). Low-dose IL-2 prevents murine chronic cardiac allograft rejection: Role for IL-2-induced T regulatory cells and exosomes with PD-L1 and CD73. Am J Transplant, 22(9), 2180-2194. https://doi.org/10.1111/ajt.17101
[58] Churlaud G, Jimenez V, Ruberte J, Amadoudji Zin M, Fourcade G, Gottrand G, et al. (2014). Sustained stimulation and expansion of Tregs by IL2 control autoimmunity without impairing immune responses to infection, vaccination and cancer. Clin Immunol, 151(2), 114-126. https://doi.org/10.1016/j.clim.2014.02.003
[59] Feng M, Guo H, Zhang C, Wang Y, Liang Z, Zhao X, et al. (2019). Absolute reduction of regulatory T cells and regulatory effect of short-term and low-dose IL-2 in polymyositis or dermatomyositis. Int Immunopharmacol, 77, 105912. https://doi.org/10.1016/j.intimp.2019.105912
[60] Rosenzwajg M, Lorenzon R, Cacoub P, Pham HP, Pitoiset F, El Soufi K, et al. (2019). Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann. Rheum. Dis., 78(2), 209-217. https://doi.org/10.1136/annrheumdis-2018-214229
[61] Fusi I, Serger C, Herzig P, Germann M, Sandholzer MT, Oelgarth N, et al. (2025). PD-1-targeted cis-delivery of an IL-2 variant induces a multifaceted antitumoral T cell response in human lung cancer. Sci Transl Med, 17(816), eadr3718. https://doi.org/10.1126/scitranslmed.adr3718
[62] Kehry MR, Yamashita LC, & Hodgkin PD. (1990). B-cell proliferation and differentiation mediated by Th-cell membranes and lymphokines. Res Immunol, 141(4-5), 421-423. https://doi.org/10.1016/0923-2494(90)90033-u
[63] Cen Z, Li Y, Wei B, Wu W, Huang Y, & Lu J. (2021). The Role of B Cells in Regulation of Th Cell Differentiation in Coxsackievirus B3-Induced Acute Myocarditis. Inflammation, 44(5), 1949-1960. https://doi.org/10.1007/s10753-021-01472-5
[64] Park E, & Ciofani M. (2025). Th17 cell pathogenicity in autoimmune disease. Exp Mol Med, 57(9), 1913-1927. https://doi.org/10.1038/s12276-025-01535-9
[65] Cheng H, Nan F, Ji N, Ma X, Zhang J, Liang H, et al. (2025). Regulatory T cell therapy promotes TGF-β and IL-6-dependent pro-inflammatory Th17 cell generation by reducing IL-2. Nat Commun, 16(1), 7644. https://doi.org/10.1038/s41467-025-62628-7
[66] Dold L, Kalthoff S, Frank L, Zhou T, Esser P, Lutz P, et al. (2024). STAT activation in regulatory CD4(+) T cells of patients with primary sclerosing cholangitis. Immun Inflamm Dis, 12(4), e1248. https://doi.org/10.1002/iid3.1248
[67] Kim S, Boehme L, Nel L, Casian A, Sangle S, Nova-Lamperti E, et al. (2022). Defective STAT5 Activation and Aberrant Expression of BCL6 in Naive CD4 T Cells Enhances Follicular Th Cell-like Differentiation in Patients with Granulomatosis with Polyangiitis. J Immunol, 208(4), 807-818. https://doi.org/10.4049/jimmunol.2001331
[68] Ballesteros-Tato A, León B, Graf BA, Moquin A, Adams PS, Lund FE, et al. (2012). Interleukin-2 inhibits germinal center formation by limiting T follicular helper cell differentiation. Immunity, 36(5), 847-856. https://doi.org/10.1016/j.immuni.2012.02.012
[69] Chaudhry A, Rudra D, Treuting P, Samstein RM, Liang Y, Kas A, et al. (2009). CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science, 326(5955), 986-991. https://doi.org/10.1126/science.1172702
[70] Rosenzwajg M, Lorenzon R, Cacoub P, Pham HP, Pitoiset F, El Soufi K, et al. (2019). Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann Rheum Dis, 78(2), 209-217. https://doi.org/10.1136/annrheumdis-2018-214229
[71] Miao M, Li Y, Xu D, Zhang R, He J, Sun X, et al. (2022). Therapeutic responses and predictors of low-dose interleukin-2 in systemic lupus erythematosus. Clin Exp Rheumatol, 40(5), 867-871. https://doi.org/10.55563/clinexprheumatol/1o6pn1
[72] Rose A, von Spee-Mayer C, Kloke L, Wu K, Kühl A, Enghard P, et al. (2019). IL-2 Therapy Diminishes Renal Inflammation and the Activity of Kidney-Infiltrating CD4+ T Cells in Murine Lupus Nephritis. Cells, 8(10). https://doi.org/10.3390/cells8101234
[73] Zhou P, Chen J, He J, Zheng T, Yunis J, Makota V, et al. (2021). Low-dose IL-2 therapy invigorates CD8+ T cells for viral control in systemic lupus erythematosus. PLoS Pathog, 17(10), e1009858. https://doi.org/10.1371/journal.ppat.1009858
[74] Kim N, Jeon YW, Nam YS, Lim JY, Im KI, Lee ES, et al. (2016). Therapeutic potential of low-dose IL-2 in a chronic GVHD patient by in vivo expansion of regulatory T cells. Cytokine, 78, 22-26. https://doi.org/10.1016/j.cyto.2015.11.020
[75] Wang F, Wang S, He B, Liu H, Wang X, Li C, et al. (2021). Immunotherapeutic strategy based on anti-OX40L and low dose of IL-2 to prolong graft survival in sensitized mice by inducing the generation of CD4(+) and CD8(+) Tregs. Int Immunopharmacol, 97, 107663. https://doi.org/10.1016/j.intimp.2021.107663
[76] Onyshchenko K, Luo R, Guffart E, Gaedicke S, Grosu AL, Firat E, et al. (2023). Expansion of circulating stem-like CD8(+) T cells by adding CD122-directed IL-2 complexes to radiation and anti-PD1 therapies in mice. Nat Commun, 14(1), 2087. https://doi.org/10.1038/s41467-023-37825-x
[77] Kalia V, Sarkar S, Subramaniam S, Haining WN, Smith KA, & Ahmed R. (2010). Prolonged interleukin-2Ralpha expression on virus-specific CD8+ T cells favors terminal-effector differentiation in vivo. Immunity, 32(1), 91-103. https://doi.org/10.1016/j.immuni.2009.11.010
[78] Shameli A, Yamanouchi J, Tsai S, Yang Y, Clemente-Casares X, Moore A, et al. (2013). IL-2 promotes the function of memory-like autoregulatory CD8+ T cells but suppresses their development via FoxP3+ Treg cells. Eur J Immunol, 43(2), 394-403. https://doi.org/10.1002/eji.201242845
[79] Li S, Xie Q, Zeng Y, Zou C, Liu X, Wu S, et al. (2014). A naturally occurring CD8(+)CD122(+) T-cell subset as a memory-like Treg family. Cell Mol Immunol, 11(4), 326-331. https://doi.org/10.1038/cmi.2014.25
[80] Horwitz DA, Zheng SG, & Gray JD. (2003). The role of the combination of IL-2 and TGF-beta or IL-10 in the generation and function of CD4+ CD25+ and CD8+ regulatory T cell subsets. J Leukoc Biol, 74(4), 471-478. https://doi.org/10.1189/jlb.0503228
[81] Minai Y, Hisatsune T, Nishijima K, Enomoto A, & Kaminogawa S. (1994). Activation via TCR or IL-2 receptor of a CD8+ suppressor T cell clone: effect on IL-10 production and on proliferation of the suppressor T cell. Cytotechnology, 14(2), 81-87. https://doi.org/10.1007/bf00758172
[82] Elrefaei M, Ventura FL, Baker CA, Clark R, Bangsberg DR, & Cao H. (2007). HIV-specific IL-10-positive CD8+ T cells suppress cytolysis and IL-2 production by CD8+ T cells. J Immunol, 178(5), 3265-3271. https://doi.org/10.4049/jimmunol.178.5.3265
[83] Grange M, Giordano M, Mas A, Roncagalli R, Firaguay G, Nunes JA, et al. (2015). Control of CD8 T cell proliferation and terminal differentiation by active STAT5 and CDKN2A/CDKN2B. Immunology, 145(4), 543-557. https://doi.org/10.1111/imm.12471
[84] Qin Y, Qian Y, Zhang J, & Liu S. (2025). CD1d-Restricted NKT Cells Promote Central Memory CD8(+) T Cell Formation via an IL-15-pSTAT5-Eomes Axis in a Pathogen-Exposed Environment. Int J Mol Sci, 26(15). https://doi.org/10.3390/ijms26157272
[85] Caligiuri MA. (2008). Human natural killer cells. Blood, 112(3), 461-469. https://doi.org/10.1182/blood-2007-09-077438
[86] Harris KE, Lorentsen KJ, Malik-Chaudhry HK, Loughlin K, Basappa HM, Hartstein S, et al. (2021). A bispecific antibody agonist of the IL-2 heterodimeric receptor preferentially promotes in vivo expansion of CD8 and NK cells. Sci Rep, 11(1), 10592. https://doi.org/10.1038/s41598-021-90096-8
[87] Hirakawa M, Matos TR, Liu H, Koreth J, Kim HT, Paul NE, et al. (2016). Low-dose IL-2 selectively activates subsets of CD4(+) Tregs and NK cells. JCI Insight, 1(18), e89278. https://doi.org/10.1172/jci.insight.89278
[88] McQuaid SL, Loughran ST, Power PA, Maguire P, Szczygiel A, & Johnson PA. (2020). Low-dose IL-2 induces CD56(bright) NK regulation of T cells via NKp44 and NKp46. Clin Exp Immunol, 200(3), 228-241. https://doi.org/10.1111/cei.13422
[89] Li Z, Lim WK, Mahesh SP, Liu B, & Nussenblatt RB. (2005). Cutting edge: in vivo blockade of human IL-2 receptor induces expansion of CD56(bright) regulatory NK cells in patients with active uveitis. J Immunol, 174(9), 5187-5191. https://doi.org/10.4049/jimmunol.174.9.5187
[90] Lee M, Bell CJM, Rubio Garcia A, Godfrey L, Pekalski M, Wicker LS, et al. (2023). CD56(bright) natural killer cells preferentially kill proliferating CD4(+) T cells. Discov Immunol, 2(1), kyad012. https://doi.org/10.1093/discim/kyad012
[91] Gumá M, Angulo A, Vilches C, Gómez-Lozano N, Malats N, & López-Botet M. (2004). Imprint of human cytomegalovirus infection on the NK cell receptor repertoire. Blood, 104(12), 3664-3671. https://doi.org/10.1182/blood-2004-05-2058
[92] Chen J, Ren E, Tao Z, Lu H, Huang Y, Li J, et al. (2025). Orchestrating T and NK cells for tumor immunotherapy via NKG2A-targeted delivery of a de novo designed IL-2Rβγ agonist. Drug Deliv, 32(1), 2482195. https://doi.org/10.1080/10717544.2025.2482195
[93] Unger ML, Hokland M, Basse PH, Nannmark U, & Johansson BR. (1997). High dose IL-2-activated murine natural killer (A-NK) cells accumulate glycogen and granules, lose cytotoxicity, and alter target cell interaction in vitro. Scand J Immunol, 45(6), 623-636. https://doi.org/10.1046/j.1365-3083.1997.d01-437.x
[94] Tiemessen MM, Jagger AL, Evans HG, van Herwijnen MJ, John S, & Taams LS. (2007). CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc Natl Acad Sci U S A, 104(49), 19446-19451. https://doi.org/10.1073/pnas.0706832104
[95] Moore KW, de Waal Malefyt R, Coffman RL, & O'Garra A. (2001). Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol, 19, 683-765. https://doi.org/10.1146/annurev.immunol.19.1.683
[96] Zhang X, Miao M, Zhang R, Liu X, Zhao X, Shao M, et al. (2022). Efficacy and safety of low-dose interleukin-2 in combination with methotrexate in patients with active rheumatoid arthritis: a randomized, double-blind, placebo-controlled phase 2 trial. Signal Transduct Target Ther, 7(1), 67. https://doi.org/10.1038/s41392-022-00887-2
[97] Xue R, Li G, Zhou Y, Wang B, Xu Y, Zhao P, et al. (2024). Efficacy and safety of low-dose interleukin 2 in the treatment of moderate-to-severe bullous pemphigoid: A single center perspective-controlled trial. J Am Acad Dermatol, 91(6), 1113-1117. https://doi.org/10.1016/j.jaad.2024.08.033
[98] Li ZG, He J, & Miao M. (2023). Low-dose interleukin-2 therapy in autoimmune and inflammatory diseases: facts and hopes. Sci. Bull., 68(1), 10-13. https://doi.org/10.1016/j.scib.2022.12.024
[99] Malek TR. (2008). The biology of interleukin-2. Annu Rev Immunol, 26, 453-479. https://doi.org/10.1146/annurev.immunol.26.021607.090357
[100] Lykhopiy V, Malviya V, Humblet-Baron S, & Schlenner SM. (2023). "IL-2 immunotherapy for targeting regulatory T cells in autoimmunity". Genes Immun, 24(5), 248-262. https://doi.org/10.1038/s41435-023-00221-y
[101] Shevach EM. (2009). Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity, 30(5), 636-645. https://doi.org/10.1016/j.immuni.2009.04.010
[102] Panduro M, Benoist C, & Mathis D. (2016). Tissue Tregs. Annu Rev Immunol, 34, 609-633. https://doi.org/10.1146/annurev-immunol-032712-095948
[103] Klatzmann D, & Abbas AK. (2015). The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat Rev Immunol, 15(5), 283-294. https://doi.org/10.1038/nri3823
[104] Castela E, Le Duff F, Butori C, Ticchioni M, Hofman P, Bahadoran P, et al. (2014). Effects of low-dose recombinant interleukin 2 to promote T-regulatory cells in alopecia areata. JAMA Dermatol, 150(7), 748-751. https://doi.org/10.1001/jamadermatol.2014.504
[105] Dong S, Hiam-Galvez KJ, Mowery CT, Herold KC, Gitelman SE, Esensten JH, et al. (2021). The effect of low-dose IL-2 and Treg adoptive cell therapy in patients with type 1 diabetes. JCI Insight, 6(18). https://doi.org/10.1172/jci.insight.147474
[106] Lim TY, Perpinan E, Londono MC, Miquel R, Ruiz P, Kurt AS, et al. (2023). Low dose interleukin-2 selectively expands circulating regulatory T cells but fails to promote liver allograft tolerance in humans. J Hepatol, 78(1), 153-164. https://doi.org/10.1016/j.jhep.2022.08.035
[107] Le Duff F, Bouaziz JD, Fontas E, Ticchioni M, Viguier M, Dereure O, et al. (2021). Low-Dose IL-2 for Treating Moderate to Severe Alopecia Areata: A 52-Week Multicenter Prospective Placebo-Controlled Study Assessing its Impact on T Regulatory Cell and NK Cell Populations. J Invest Dermatol, 141(4), 933-936.e936. https://doi.org/10.1016/j.jid.2020.08.015
[108] Humrich JY, Cacoub P, Rosenzwajg M, Pitoiset F, Pham HP, Guidoux J, et al. (2022). Low-dose interleukin-2 therapy in active systemic lupus erythematosus (LUPIL-2): a multicentre, double-blind, randomised and placebo-controlled phase II trial. Ann. Rheum. Dis., 81(12), 1685-1694. https://doi.org/10.1136/ard-2022-222501
[109] Koreth J, Kim HT, Jones KT, Lange PB, Reynolds CG, Chammas MJ, et al. (2016). Efficacy, durability, and response predictors of low-dose interleukin-2 therapy for chronic graft-versus-host disease. Blood, 128(1), 130-137. https://doi.org/10.1182/blood-2016-02-702852
[110] Farooq A, Trehan S, Singh G, Arora N, Mehta T, Jain P, et al. (2024). A Comprehensive Review of Low-Dose Interleukin-2 (IL-2) Therapy for Systemic Lupus Erythematosus: Mechanisms, Efficacy, and Clinical Applications. Cureus, 16(9), e68748. https://doi.org/10.7759/cureus.68748
[111] Malek TR, & Bayer AL. (2004). Tolerance, not immunity, crucially depends on IL-2. Nat Rev Immunol, 4(9), 665-674. https://doi.org/10.1038/nri1435
[112] Todd JA, Evangelou M, Cutler AJ, Pekalski ML, Walker NM, Stevens HE, et al. (2016). Regulatory T Cell Responses in Participants with Type 1 Diabetes after a Single Dose of Interleukin-2: A Non-Randomised, Open Label, Adaptive Dose-Finding Trial. PLoS Med, 13(10), e1002139. https://doi.org/10.1371/journal.pmed.1002139
[113] Miao M, Xiao X, Tian J, Zhufeng Y, Feng R, Zhang R, et al. (2021). Therapeutic potential of targeting Tfr/Tfh cell balance by low-dose-IL-2 in active SLE: a post hoc analysis from a double-blind RCT study. Arthritis Res. Ther., 23(1), 167. https://doi.org/10.1186/s13075-021-02535-6
[114] He J, Zhang X, Wei Y, Sun X, Chen Y, Deng J, et al. (2016). Low-dose interleukin-2 treatment selectively modulates CD4(+) T cell subsets in patients with systemic lupus erythematosus. Nat. Med., 22(9), 991-993. https://doi.org/10.1038/nm.4148
[115] Humrich JY, & Riemekasten G. (2019). Low-dose interleukin-2 therapy for the treatment of systemic lupus erythematosus. Curr Opin Rheumatol, 31(2), 208-212. https://doi.org/10.1097/bor.0000000000000575
[116] von Spee-Mayer C, Siegert E, Abdirama D, Rose A, Klaus A, Alexander T, et al. (2016). Low-dose interleukin-2 selectively corrects regulatory T cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis., 75(7), 1407-1415. https://doi.org/10.1136/annrheumdis-2015-207776
[117] Hu FY, Wang J, Zhang SX, Su R, Yan N, Gao C, et al. (2021). Absolute reduction of peripheral regulatory T cell in patients with relapsing polychondritis. Clin Exp Rheumatol, 39(3), 487-493. https://doi.org/10.55563/clinexprheumatol/qndtvt
[118] Rosenzwajg M, Salet R, Lorenzon R, Tchitchek N, Roux A, Bernard C, et al. (2020). Low-dose IL-2 in children with recently diagnosed type 1 diabetes: a Phase I/II randomised, double-blind, placebo-controlled, dose-finding study. Diabetologia, 63(9), 1808-1821. https://doi.org/10.1007/s00125-020-05200-w
[119] Saadoun D, Rosenzwajg M, Joly F, Six A, Carrat F, Thibault V, et al. (2011). Regulatory T-cell responses to low-dose interleukin-2 in HCV-induced vasculitis. N. Engl. J. Med., 365(22), 2067-2077. https://doi.org/10.1056/NEJMoa1105143
[120] Wang D, Fu B, Shen X, Guo C, Liu Y, Zhang J, et al. (2021). Restoration of HBV-specific CD8(+) T-cell responses by sequential low-dose IL-2 treatment in non-responder patients after IFN-alpha therapy. Signal Transduct Target Ther, 6(1), 376. https://doi.org/10.1038/s41392-021-00776-0
[121] Zhou X, Wang Y, Huang B, Feng R, Zhou X, Li C, et al. (2023). Dynamics of T follicular helper cells in patients with rheumatic diseases and subsequent antibody responses in a three-dose immunization regimen of CoronaVac. Immunology, 168(1), 184-197. https://doi.org/10.1111/imm.13572
[122] Zhang B, Sun J, Wang Y, Ji D, Yuan Y, Li S, et al. (2021). Site-specific PEGylation of interleukin-2 enhances immunosuppression via the sustained activation of regulatory T cells. Nat. Biomed. Eng., 5(11), 1288-1305. https://doi.org/10.1038/s41551-021-00797-8
[123] Levin AM, Bates DL, Ring AM, Krieg C, Lin JT, Su L, et al. (2012). Exploiting a natural conformational switch to engineer an interleukin-2 'superkine'. Nature, 484(7395), 529-533. https://doi.org/10.1038/nature10975
[124] Sockolosky JT, Trotta E, Parisi G, Picton L, Su LL, Le AC, et al. (2018). Selective targeting of engineered T cells using orthogonal IL-2 cytokine-receptor complexes. Science, 359(6379), 1037-1042. https://doi.org/10.1126/science.aar3246
[125] Horwitz DA, Kim D, Kang C, Brion K, Bickerton S, & La Cava A. (2025). CD2-targeted nanoparticles encapsulating IL-2 induce tolerogenic Tregs and TGF-β-producing NK cells that stabilize Tregs for long-term therapeutic efficacy in immune-mediated disorders. Front Immunol, 16, 1587237. https://doi.org/10.3389/fimmu.2025.1587237
[126] Koh B, Oz STG, Sato R, Nguyen HN, Dunlap G, Mahony C, et al. (2025). Functional and dysfunctional T regulatory cell states in human tissues in RA and other autoimmune arthritic diseases. bioRxiv, 10.1101/2025.09.23.677874. https://doi.org/10.1101/2025.09.23.677874
[127] Raposo A, Paço S, Ângelo-Dias M, Rosmaninho P, Almeida ARM, & Sousa AE. (2025). The distinctive signature of regulatory CD4 T cells committed in the human thymus. Front Immunol, 16, 1553554. https://doi.org/10.3389/fimmu.2025.1553554
[128] Cheng Z, Wang LJ, Honaker Y, Cincotta SA, Page CE, Vollhardt S, et al. (2025). Transcriptional fingerprinting of regulatory T cells: ensuring quality in cell therapy applications. Front Immunol, 16, 1602172. https://doi.org/10.3389/fimmu.2025.1602172
[129] Arroyo-Olarte RD, Flores-Castelán JC, Armas-López L, Escobedo G, Terrazas LI, Ávila-Moreno F, et al. (2024). Targeted Demethylation of FOXP3-TSDR Enhances the Suppressive Capacity of STAT6-deficient Inducible T Regulatory Cells. Inflammation, 47(6), 2159-2172. https://doi.org/10.1007/s10753-024-02031-4
[130] Kressler C, Gasparoni G, Nordström K, Hamo D, Salhab A, Dimitropoulos C, et al. (2020). Targeted De-Methylation of the FOXP3-TSDR Is Sufficient to Induce Physiological FOXP3 Expression but Not a Functional Treg Phenotype. Front Immunol, 11, 609891. https://doi.org/10.3389/fimmu.2020.609891
[131] Xu K, Liu Q, Wu K, Liu L, Zhao M, Yang H, et al. (2020). Extracellular vesicles as potential biomarkers and therapeutic approaches in autoimmune diseases. J Transl Med, 18(1), 432. https://doi.org/10.1186/s12967-020-02609-0
[132] Feng R, Xiao X, Huang B, Zhang K, Zhang X, Li Z, et al. (2024). Predictive biomarkers for low-dose IL-2 therapy efficacy in systemic lupus erythematosus: a clinical analysis. Arthritis Res Ther, 26(1), 180. https://doi.org/10.1186/s13075-024-03388-5
[133] Friedberg JW, Neuberg D, Gribben JG, Fisher DC, Canning C, Koval M, et al. (2002). Combination immunotherapy with rituximab and interleukin 2 in patients with relapsed or refractory follicular non-Hodgkin's lymphoma. Br J Haematol, 117(4), 828-834. https://doi.org/10.1046/j.1365-2141.2002.03535.x
[134] Zhou H, Zhao X, Zhang R, Miao M, Pei W, Li Z, et al. (2023). Low-dose IL-2 mitigates glucocorticoid-induced Treg impairment and promotes improvement of SLE. Signal Transduction and Targeted Therapy, 8(1). https://doi.org/10.1038/s41392-023-01350-6
[135] Cases M, Ritter N, Rincon-Arevalo H, Kroh S, Adam A, Kirchner M, et al. (2025). Novel non-coding FOXP3 transcript isoform associated to potential transcriptional interference in human regulatory T cells. RNA Biol, 22(1), 1-20. https://doi.org/10.1080/15476286.2025.2502719
[136] Ramón-Vázquez A, Flood P, Cashman TL, Patil P, & Ghosh S. (2025). T lymphocyte plasticity in chronic inflammatory diseases: The emerging role of the Ikaros family as a key Th17-Treg switch. Autoimmun Rev, 24(3), 103735. https://doi.org/10.1016/j.autrev.2024.103735
[137] Lu Y, & Man XY. (2025). Diversity and function of regulatory T cells in health and autoimmune diseases. J Autoimmun, 151, 103357. https://doi.org/10.1016/j.jaut.2025.103357
[138] Bothur E, Raifer H, Haftmann C, Stittrich AB, Brüstle A, Brenner D, et al. (2015). Antigen receptor-mediated depletion of FOXP3 in induced regulatory T-lymphocytes via PTPN2 and FOXO1. Nat Commun, 6, 8576. https://doi.org/10.1038/ncomms9576
[139] Long SA, Cerosaletti K, Wan JY, Ho JC, Tatum M, Wei S, et al. (2011). An autoimmune-associated variant in PTPN2 reveals an impairment of IL-2R signaling in CD4(+) T cells. Genes Immun, 12(2), 116-125. https://doi.org/10.1038/gene.2010.54
[140] Lin JX, Ge M, Liu CY, Holewinski R, Andresson T, Yu ZX, et al. (2024). Tyrosine phosphorylation of both STAT5A and STAT5B is necessary for maximal IL-2 signaling and T cell proliferation. Nat Commun, 15(1), 7372. https://doi.org/10.1038/s41467-024-50925-6
[141] Santarlasci V, Mazzoni A, Capone M, Rossi MC, Maggi L, Montaini G, et al. (2017). Musculin inhibits human T-helper 17 cell response to interleukin 2 by controlling STAT5B activity. Eur J Immunol, 47(9), 1427-1442. https://doi.org/10.1002/eji.201746996
[142] Li Y, Lin D, Chen J, Zhang W, Jin H, Feng S, et al. (2025). Alkaline phosphatase-responsive hydrogel for efficient management of autoimmune intraocular inflammation. J Control Release, 387, 114229. https://doi.org/10.1016/j.jconrel.2025.114229
[143] Wahbi M, Wen Y, Kontopoulou M, & De France KJ. (2025). Modification of cellulose nanocrystals with epoxidized canola oil for enhancing interfacial compatibility with poly(lactic acid). Carbohydr Polym, 370, 124471. https://doi.org/10.1016/j.carbpol.2025.124471
[144] Shin H, Kang S, Won C, & Min DH. (2023). Enhanced Local Delivery of Engineered IL-2 mRNA by Porous Silica Nanoparticles to Promote Effective Antitumor Immunity. ACS Nano, 17(17), 17554-17567. https://doi.org/10.1021/acsnano.3c06733
[145] Jeon EY, Choi DS, Choi S, Won JY, Jo Y, Kim HB, et al. (2023). Enhancing adoptive T-cell therapy with fucoidan-based IL-2 delivery microcapsules. Bioeng Transl Med, 8(1), e10362. https://doi.org/10.1002/btm2.10362
[146] Wang J, Zhou L, & Liu B. (2020). Update on disease pathogenesis, diagnosis, and management of primary Sjögren's syndrome. Int J Rheum Dis, 23(6), 723-727. https://doi.org/10.1111/1756-185x.13839
[147] Streckfus C, Bigler L, Navazesh M, & Al-Hashimi I. (2001). Cytokine concentrations in stimulated whole saliva among patients with primary Sjögren's syndrome, secondary Sjögren's syndrome, and patients with primary Sjögren's syndrome receiving varying doses of interferon for symptomatic treatment of the condition: a preliminary study. Clin Oral Investig, 5(2), 133-135. https://doi.org/10.1007/s007840100104
[148] Zhao Z, Zhang X, Su L, Xu L, Zheng Y, & Sun J. (2018). Fine tuning subsets of CD4(+) T cells by low-dosage of IL-2 and a new therapeutic strategy for autoimmune diseases. Int Immunopharmacol, 56, 269-276. https://doi.org/10.1016/j.intimp.2018.01.042
[149] Villani AC, Sarkizova S, & Hacohen N. (2018). Systems Immunology: Learning the Rules of the Immune System. Annu Rev Immunol, 36, 813-842. https://doi.org/10.1146/annurev-immunol-042617-053035
[150] Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly YM, et al. (2013). The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science, 341(6145), 569-573. https://doi.org/10.1126/science.1241165
[151] Verra N, Jansen R, Groenewegen G, Mallo H, Kersten MJ, Bex A, et al. (2003). Immunotherapy with concurrent subcutaneous GM-CSF, low-dose IL-2 and IFN-alpha in patients with progressive metastatic renal cell carcinoma. Br J Cancer, 88(9), 1346-1351. https://doi.org/10.1038/sj.bjc.6600915
Type
Published
Data Availability Statement
All data needed to evaluate the conclusions in the paper are present in the paper. Additional data related to this paper may be requested from the authors.
Issue
Section
License
Copyright (c) 2026 Life Conflux

This work is licensed under a Creative Commons Attribution 4.0 International License.







